Document Type : Original Article
Authors
Department of Food Sciences, College of Sciences Agriculture Engineering Baghdad University, Baghdad, Iraq
Abstract
Casein is a biologically and physiologically complex fluid that contains proteins, water, fat, lactose, minerals, and vitamins as its principal constituents that might support the treatment of human body. Isolation of casein using urea and salts was used. Then, casein was partially purified using DEAE-Cellulose and Sephadex-G-75. The hydrolysis degree was estimated after incubation with pepsin, trypsin, and a mixture of them. αs-CN was partially purified with one peak after DEAE-cellulose ion exchange and there is also one peak that appeared after the Sephadex G-25 gel filtration technique. The maximum hydrolysis was gained after 8 hours of incubation with hydrolyzing enzymes. αH hydrolysates of αs-casein showed that the inhibition rate reached about 67% after 8 hours of incubation with a mixture of pepsin and trypsin with a hydrolysis concentration of 0.114 μmol. Results showed that casein extracted from sheep milk can be highly purified using both ion exchange and gel filtration chromatography. Hydrolysis of αs-CN produces low molecular weight protein using pepsin and trypsin and a mixture of them. The αs-CN has a high inhibition rate of the angiotensin-converting enzyme (ACE).
Graphical Abstract
Keywords
Main Subjects
Introduction
Milk has positive effects on many body structures as the nervous, immune, and cardiovascular systems [1]. Furthermore, milk is representing the core of a lot of bio-active peptides with major immunological and nutritive activities [2]. A lot of bio-active peptides are used for the treatment of many diseases such as mineral malabsorption, hypertension, diarrhea, dental health diseases, thrombosis, and diseases related to immune disorders. The casein (CN) contains: calcium, mineral levels, and phosphate ratios in the skim milk varied between species, but no differences in the Calcium: Phosphor ratios in the sera and casein micelles were found [3].
The peptide article field the most serious article fields of preventative clinical chemistry when there has been a clinical guide that the lack of peptides resulted in the effectiveness of reduced blood pressure, and hypotension which led to the serious concern that the impact resulted from ACE1 inhibition only as biochemical drugs or the impact of inhibition enzymes and the answer was “almost with an obvious and direct and impact on ACE1 [4]. On a global level, there are a lot of products containing hypotensive peptides in the markets and pharmaceutical formulations, especially seaweed peptides, sour milk, and peanuts. All products, if not all, inhibit the activity of ACE, and other functions of animal organs and humans, most antimicrobial and antioxidant peptide, these characteristics are found in the bio-active peptides [5].
Caseins are essentially composed of 4 different types, namely the αs1, αs2, κ, and β-CN [6]. The characteristics of the milk casein (CN) are different among the species, and the micelle properties also differ with regard to hydration, size, and mineralization.
Milk casein (CN) micelles in sheep have a high mineralization score and are heat stable and less hydrated [7].
Milk protein is a rich protein that is released with special hypotensive peptides which can have more than 1 bio-active function type of these peptides rely on the characteristic of protein type and their primary structure, which determine the resistance level to the protein proteolysis enzyme, especially pepsin and trypsin enzymes that work in the digestive tract [8].
The biological activity of low molecular weight molecules is based on the sequence and type of the amino acids forming the peptide. Bio-active peptides almost contain a range from 3 to 20 amino acids per 1 molecule [9].
Purification and hydrolysis protein of casein from sheep milk to produce peptides with inhibitory effects on ACE1 that are rare and limited [10] reported that rare study about ACE-inhibition from proteolytic hydrolysate derived from casein (CN) sheep milk, the experiment revealed that casein (CN) hydrolyzed by trypsin and pepsin with increased ACE1 inhibition efficacy which equals 67.1 and this activity increases with the hydrolysis degree that was 48.0 [10]. Hence, the peptides isolation from their hydrolysates leads to a raise in the ACE1 inhibition rate% to 97.6%. Val-Pro-Pro is the most successful peptides that resist digestive enzymes and have an ACE1 inhibition rate. Therefore, it can be consumed orally. This study aimed to extract and purify αs-CN from sheep milk to produce low molecular weight hydrolysate that is able to inhibit ACE1 [10].
Materials and Methods
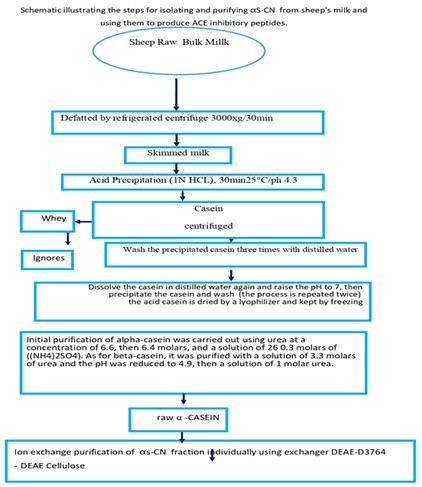
αS-CN purification
The method was utilizied to purify αs-CN process from milk with modification in acidic casein quantity that utiliezed at the beginning of the separation process as 40 g of acidic casein prepared in 100 mL of urea solution (6.6 M) with stirring. Distilled water (42.5 mL) was added to the solution to gain 4.63 M of urea solution and preserved at 25 °C until it form white precipitate which dissolved in solution of urea (100 mL) containing 0.421 M NaCl and decrease urea concentration to 5.11 M, the αs-CN precipitates have kept in freeze drying [11].
Purification of αs-casein using Ion exchange chromatography
Crude αs-CN sample was purified by Anion exchange chromatography from the precipitates according to Whitney, (1988) [12]. 1 g of freeze-dried αs-CN has been dissolved in NaPO4 buffer (100 mL), (0.1 M), and pH 7.4 that include urea (3.3 M) and mercaptoethanol (0.010 M) then mix with DEAE-cellulose material for 15 min. DEAE-Cellulose exchange mix was poured into a column with dimensions of (2×50 cm). The column was washed with buffer solution to conjugate caseins with the column and then eluted to extract αs-casein [13].
The αs-CN sample was removed from a column using phosphate buffer used to dissolve casein containing NaCl in gradient concentrations of 0.1, 0.175, 0.2, and 0.25 M. The flow rate was 50 mL/hour and 5 mL/tube by using fraction collector protein solutions in the tube was evaluated by spectrophotometer device at 280 nm (Shimadzu model 1650 PC).
Purification of αs-casein by gel filtration technique
Sephadex G-75 gel is utilized to prepare gel filtration columns according to Pharmacia’s fine chemicals firm. Freeze-dried αs-CN (0.5 g) were purified by the DEAE-Cellulose chromatography in 10 mL, 0.005 M phosphate buffer. Sephadex G-75 with column dimension of (90×1.5 cm) balanced by the same buffer sodium chloride including urea (6.6 M) and EDTA (0.004 M). The eluted fractions from Sephadex G-75 column of 20 mL/hr flow rate and 3 mL/tube. Protein solutions in tubes read by Shimadzu model 1650 PC spectrophotometer at 280 nm. The αs-CN total volume and protein concentration was estimated by the Bradford method.
Protein concentration assay
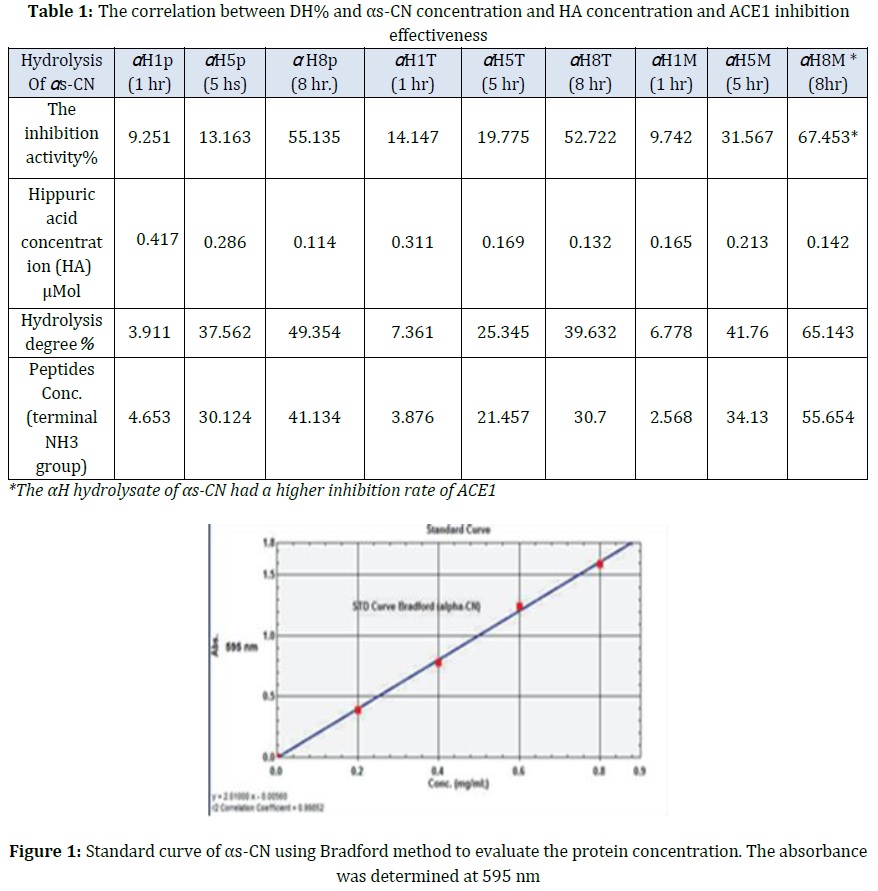
Bradford method utiliezed to estimate αs-CN concentration in solutions using standard αs-CN in standard curve preparation as reported in Table 1 to determine the purity and molecular weight estimation of αs-CN (Figure 1). To identify the αs-casein purity and estimate its molecular weight, the polyacrylamide gel electrophoresis method was utilized in the presence of sodium dodecyl sulfate via a vertical electrophoresis device compared with standard bovine and known molecular weight αs-CN based on the Laemmli, (1970) described method and modified by Schägger (2006) [14].
The hydrolysis of αs-Casein (CN) by proteolytic enzymes
The method that followed by the modifications of (10) in the protein amount and the enzymatic proteolytic units number in proportion to the amount of protein gained from all purification steps [15].
0.1 g of αs-CN was dissolved in 10 mL D.W with pH 2 for pepsin and pH 8 for trypsin then raised to pH 8 for a mix of trypsin and pepsin at a 1:1 ratio. The change of pH by adding both NaOH and HCl. Trypsin and Pepsin were added to the reaction mix as 20, and 18 units respectively, within 8 hours of incubation at room temperature. The time of reaction enzymes was 4 hours for both trypsin and pepsin. Samples were pulled for hours to evaluate the hydrolysis degree in αs-CN.
Estimation of degree hydrolysis
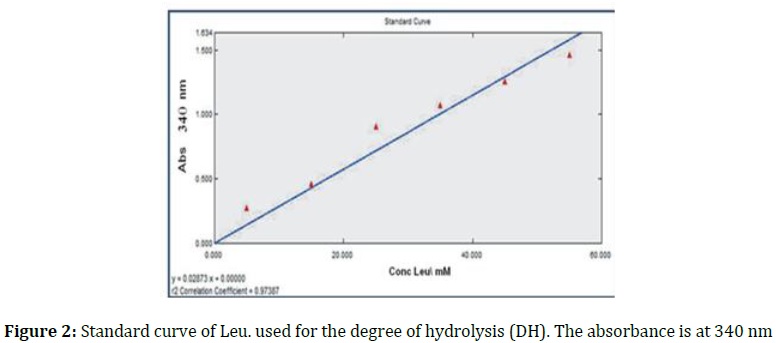
The DH is evaluated according to the literature [16]. Figure 2 showed that the standard curve of leucine is utilized to determine the casein DH. The 0.25 mL was delivered to a test tube each time and mixed with NaPO4 buffer (0.235 M) with pH=8.2. After that, 2 mL of Tri-nitrobenzene sulfuric acid (0.1%) was added to the reactive mixture and incubated for 1 hour at 50 °C. After that, it was isolated from light and stopped reaction by adding HCl (0.1 M) and left to cool to 30 ± 2 °C for 30 min. The absorbance was determined at 340 nm. The DH (Peripheral NH3 groups or peptides concentration) was determined by entering the equation below using the UV-PROBE 2.1 program that utilizes Shimadzu devices SP:
![]()
So that,
Lt= The Peripheral NH3 group conc. at (0-8 hr.).
L0 = the Free a.a in the original protein samples without treatment.
Lmax = L total
Lmax = The total free a.a that resulted from acidic hydrolysis using 6 M HCl, 120 cp for 24 hours.
Results and Discussion
Acidic αs-CN preparation and composition
Milk samples from sheep were collected in December 2021 from the field in the College of Agriculture, University of Baghdad. Results in Table 1 revealed the sheep milk moisture was 83.2% and the solid non-fat was 10.40%. It also revealed that the total solid in sheep milk was (16.8%), Furthermore, it revealed that the protein level% in local sheep milk was 5.3%, these results agree with the standard value of total solid in sheep milk (15-20%) as mentioned by park et al. (2007) [16]. The other studies also higher levels of total solids (19.11%), fat (7.52%), lactose (4.55%), protein (5.90%), and solids-non-fat (11.45%) [17]. The sheep milk composition showed an increased level of fat (6.4%). These values are considered very close to fat% in sheep milk as mentioned by [18].
Carbohydrate content in sheep milk has been identified (4.2%), which was within the normal range of carbohydrates in sheep milk (4.2%) in sheep milk [19]. The Ash ratio in local sheep milk was 0.91% which is higher than the ash ratio in cow milk (0.7%). The acidity and pH of milk were estimated in this study (0.23 % and 6.61), respectively. The acidity of sheep milk is higher than cow milk due to the higher total solid in sheep milk (20), while a higher acidity (9.3), and pH value (6.77) were indicated in another study [17].
The density and refractive index for sheep milk were 1.045 g/cm3 and 1.354 g/cm3 at 20 °C. These values were higher than the density and refractive index range (1.0347-1.0384 g/cm3) and (1.3492-1.3497 g/cm3) which might be related to sheep breed difference [17].
Extraction of crude acidic casein from sheep milk
Crude Acidic Casein was extracted from local sheep milk using acidic precipitation followed by the cryogenic centrifugation method. The results showed that the total amount of protein in milk sheep was 5.86 g/100 g (57%), while the total amount of acidic casein protein was 4.5 g/100 g (43%), which was within the normal range of the total amount of acidic casein protein in sheep milk (4.2-5.2 g/100 g). These results agree with [20, 21] which showed that the minimum protein and casein amount in sheep milk was 5.5 and 4.7 g/100 g, respectively, for many breeds. The protein portion has a huge impact on the technological and nutritional value of milk.
Purification of primary crude αs-casein in sheep milk
The initial separation of total alpha-casein before purification methods were carried out by chromatographic techniques [22]. The results indicated in this study showed that the primary weight of total acidic casein used to precipitate αs-casein using salts and urea that was 16 g per 40 g. These agree with some research that used 30 g of acidic casein to precipitate 9 g [11]. Studies revealed that the normal standard weight of αs-casein in sheep milk was 15-22 g, which is similar to our study. It showed that αs-casein was 6.7% and 22.8%, respectively. These results are inconsistent with [23] who showed that the obtained αs-CN% was 15%, using the sedimentation method with urea and salts, while [21] disagree with these results, it reported that 29% of αs-CN was obtained due to the presence of two types of αs-CN, in small quantities in the models [21].
Purification of αs-Casein in sheep milk by anion exchanger-DEAE-Cellulose
αs-CN isolated from sheep milk, and then purified by Ion-Exchange chromatography technique using DEAE-Cellulose an anionic exchanger. Results in Figures 3 revealed that one protein peak after the washing step, while there were different protein peaks was appeared after the elution step with gradient concentrations of NaCl solution. The protein peaks in the washing and elution steps were detected by measuring the absorbance at 280 nm for each eluted fraction. The eluted proteins were assayed to detect αs-CN concentration.
Results illustrated in Figures 3 indicated that there was only one eluted peak in fraction number 15 to 35 using (0.5 M) sodum chloridesolution, and it belongs to αs-CN due to it has a strong negative charge that was recovered from the column when using a (0.5 M) NaCl solution due to the nature of its constituent acids, the two amino acids aspartic and glutamic, form the highest proportions of the rest of the acids in the αs -CN molecule.
Fractions representing αs-CN were collected and pooled, then protein concentrations were estimated. The amount of the eluted αs-CN was 125 ml used for the next purification step. The protein concentration was estimated (16.7 g/mL). These results disagree with [23] who showed that there is no peak in the wash step, while there are four peaks that appeared after separation with the DEAE-Cellulose anion exchanger.
Purification of αs-casein by gel filtration technique using Sephadex G-75 column
Sephadex G-75 gel was used to make the gel filtration column, according to the method used by [22] in purified αs-CN with a modification in column length only, as the column was based on the molecular weight differences between the proteins [24]. Results in Figure 4 showed that only 1 peak representing αs-CN appeared after elution with potassium phosphate buffer. Fractions represent αs-CN pooled, and then protein concentration was estimated in using the Bradford method. The results agree with [25] and [26] who confirmed the possibility of obtaining one major protein peak for casein fractions of cow, sheep, and goat milk by the ion exchange method, and then gel filtration with a Sephadex column.

Purification of αs-CN using SDS-page
The purity and molecular weight of αs-CN, were determined using the SDS-PAGE method which depends on the electrophoresis of protein according to their charge. The results indicated in Figure 5 showed that there is single-purified αs-CN band appeared after electrophoresis of αs-CN which confirms that it is free of other casein fractions, the purified αs-CN has migrated at the same distance as standard proteins, which confirmed the purity of the protein. αs-CN is predominant in sheep milk casein, as indicated by [27] and [28]. The molecular weight of pure αs-CN was 25.620 KD and 26 KD, respectively, compared with the molecular weight of the standard protein in Ladder. The results agree with [29] and [30] who found that αs-CN was migrated to less than 26.8 KD in SDS-PAGE.
Hydrolysis of αs-Casein
Hydrolysis of αs-casein from sheep milk was evaluated using Pepsin and Trypsin and a mix of both Enzymes for eight hours [32, 33]. The results are shown in Figure 6 reveal that the hydrolysis degree increased after the incubation of αs-CN with trypsin and pepsin due to high absorption values at 340 nm wavelength. This means an enhanced release of peptides due to the specific hydrolysis action of the pepsin and trypsin enzymes and the whole behavior of the 3 treatments is similar in that they reached the optimum hydrolysis after 8 hours of incubation at 37 °C but in different percentages 58.2, 51.1, and 66.1% with pepsin, trypsin and their mixture, respectively [35].
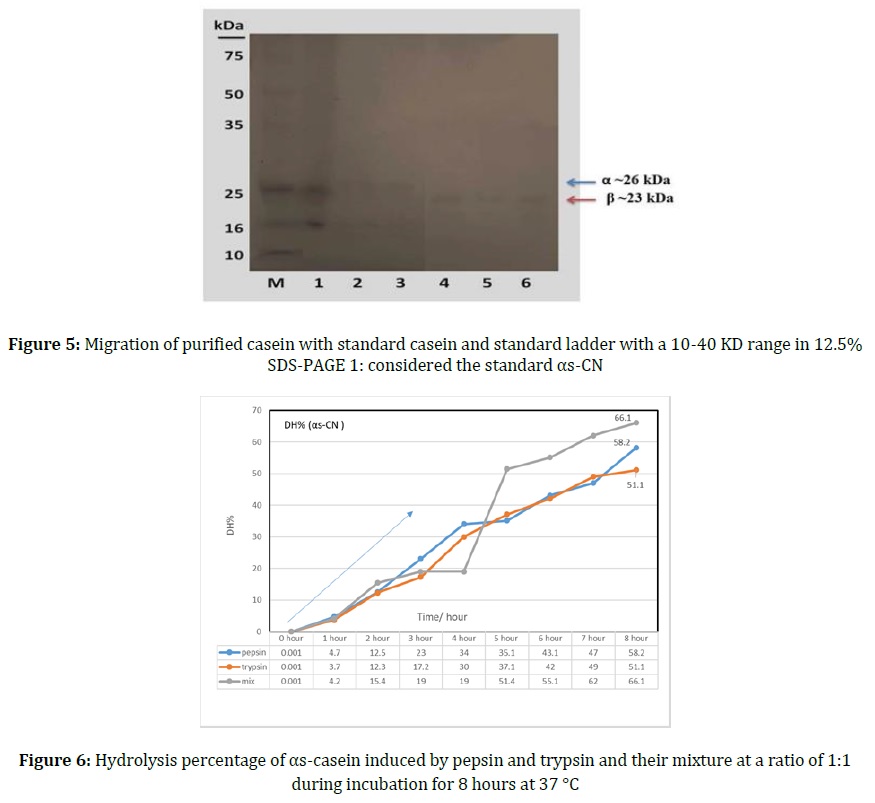
As a result of the synergistic activity of the pepsin and trypsin enzymes together on the DH of αs-CN, the results in Figure 6, showed that there is an increase in DH of αs-CN after 4 hours of incubation with the pepsin enzyme (34.0%) which increased after 4 hours of incubation with trypsin to 30.0%.
Results in Figure 6 demonstrated that the optimum level of DH was obtained after incubation with a mixture of trypsin and pepsin after eight hours of incubation. These results are inconsistent with [23] which showed that DH enhanced to the highest level after incubation of pepsin and trypsin together to hydrolyze α-CN.
Trypsin was used in the hydrolysis immediately after different proteolytic enzymes to increase the DH of casein from 44.5 to 72%. The high hydrolysis rates after purification of casein from sheep milk indicated that αs-CN and β-CN have low molecular weight peptides obtained after hydrolysis according to the structure of αs-CN, their biochemical components of amino acids and sequences, in addition to the type and concentration of enzymes used in the analysis, and the increase of αs-casein decomposers by pepsin and trypsin in sheep [30].
The inhibitory effects of αs-CN on angiotensin-converting enzyme (ACE1) Hydrolysates
The inhibiting activity of the hydrolysates produced by the hydrolysis of alpha- and beta-casein was estimated, which was termed αs-CN for three different periods (1, 5, and 8) hours of hydrolysis with pepsin, trypsin, and their (1:1) ratio mixture.
The inhibitory effectiveness of ACE1 by αs – CN hydrolysates (αH)
Results indicated in Figure 7 and Table 1 showed three hydrolysates after determining the inhibitory effect of αs -CN Hydrolysates (αH) on ACE1. The αs -CN Hydrolysates hydrolyzed by the activity of pepsin were incubated with ACE1 for three different periods (1, 5, and 8 hours) referred to as αH1P, αH5P, and αH8P. In the same method, results also showed three hydrolysates by trypsinization of αs -CN hydrolysates referred to as αH1P, αH5P, and αH8P in addition to three hydrolyses by the effect of a mixture of pepsin: trypsin after incubation for three different interval times (1, 5, and 8 hours) on ACE1 activity. The ACE1 activity is determined by the measurement of HA concentration produced by the enzymatic activity of ACE1 on the hypuric acid substrate (HHL). The results showed an inverse proportion between HA and the inhibition%, as depicted in Figure 7. However, the results pointed out that the inhibition% was 50% higher in hydrolysate produced after 8 hours of incubation with pepsin, trypsin, and their mixture (52.722%, 55.135%, and 67.453%, respectively) than the other hydrolysate produced after 1 and 5 hours of incubation with the same hydrolyzing enzymes with the following concentration, respectively (0.114, 0.132, and 0.142 µMol).
Results also showed that the increased levels of DH led to an increase in the inhibitory activity of the hydrolysates, which resulted in αs-CN hydrolysis, whether it was a group of peptides or only one peptide chain. Results in Table 1 and Figure 7 pointed out that there is an increase in the inhibition rate of ACE1 during the period between 5 hours and 8 hours. While it showed that there is a lower inhibition rate after incubation for 1 hour and up to 5 hours. The αH8M with the highest inhibition rate of ACE1 (67.453%) was collected for more purification processes to identify the peptide that is responsible for the inhibitory activity within the hydrolysate, as indicated in Table 1.
The active peptides block the transformation process of HHL to HA, the theory was confirmed by many studies to demonstrate the hydrolytic effect of the ACE1 activity. It found that casein was hydrolyzed using pepsin and trypsin to obtain an inhibitory activity that reached 72% after 6 hours of incubation and the peptide concentration was 60.06 mM [31]. These theories also agree with (27); and (28) who showed that DH% of sheep and goats milk degraded to more than 50% after increasing the hours of incubation with trypsin and pepsin. On the other hand, some researchers indicated that the increase in DH% is not the only factor that increases the efficiency of ACE1 inhibition, especially in the first hours of incubation.
However, it was the peptide concentration and the increase in the peptide types especially at using different types of inhibitory enzymes, the diversity of enzymes used in the study might increase the activation of catalyst sites of ACE1 and will block the binding of an enzyme with its substrate in the human body [34].

Conclusion
It is concluded that αs-CN can be highly purified using DEAE-Cellulose ion exchange and the Sephadex G-75 chromatography method. The molecular weight of αs-CN has low molecular weight. The synergistic action between trypsin and pepsin will obtain a high degree of hydrolysis of αs-CN from sheep milk and produce a higher inhibition rate of ACE1 after 8 hours of incubation.
Acknowledgements
The authors present deep gratitude to Baghdad University, College of Agriculture for the support of this research.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
Conflict of Interest
We have no conflicts of interest to disclose.
ORCID
Shatha Jameel AlKhalidy
https://orcid.org/0000-0002-3565-3015
HOW TO CITE THIS ARTICLE
Shatha Jameel AlKhalidy, kifah saed Dosh. ACE1-Inhibiting Activity of αs-Casien Isolated from Sheep Milk. Chem. Methodol., 2023, 7(2) 156-167