Document Type : Original Article
Authors
Department of Chemistry, College of Education for Pure Science Ibn Al-Haitham, University of Baghdad, Iraq
Abstract
The new compounds of 3-substituted phenyl-5-(3,4,5-trihydroxyphenyl)-4H-pyrazole-4-carbaldehyde [III]a-e were synthesized by reacting various hydrazones derived from galloyl hydrazide with different substituted aromatic ketones using phosphoryl trichloride in dimethylformamide as a solvent. The newly synthesized derivatives were elucidated using FT-IR, 1H-NMR and mass spectroscopy. The antimicrobial activity of these derivatives was examined using two types of pathogenic bacteria and most of the derivatives exhibited excellent and good efficacy contra these species of bacteria using ampicillin as standard.
Graphical Abstract
Keywords
Main Subjects
Introduction
Pyrazole ring is the important types of heterocyclic compounds show a broad spectrum in medicine drugs, it is an aromatic five-membered diazoles including two nitrogen and heteroatoms. Various pyrazole derivatives have been founder their application as non-steroidal anti-inflammatory drugs, like antipyrine, aminopyrine, and oxyphenbutazone [1-4]. On the other hand, many pyrazole-4-carbaldehyde derivatives exhibited anti-bacterial, anti-cancer, and anti-parasitic activities. Aromatic diazole carboxaldehyde is activated completely for undergo Vilsmeier-Haack reactions in the accepted subject to give 4-formylpyrazole. Likewise, hydrazone can be cyclized and go through Vilsmeier-Haack reaction to give 4-formylpyrazole. Formyl group can be used efficiently in the synthesis of heterocyclic compounds inserted in organic substrates by mild reagent such as Vilsmeier-Haack reaction [5-8]. Biological potential of the structural formula of galloyl hydrazide is a significant species unit in synthetic pharmaceuticals has encouraged the development of therapeutic potential; it has also been used as potent precursors for bioactive compounds [9-11]. In present work, an attempt was to synthesis 3-aryl substituted pyrazole-4-carbaldehyde derivatives and evaluation of their antimicrobial efficiency against some microorganisms.
Materials and Methods
Multiple devices were used for characterization of the synthesized compounds, among these techniques: FT-IR (8300s) Shimadzu with KBr disc, (1H- and 13C-NMR Spectra), Bruker (Ultra Shield 500 MHz) (in DMSO-d6 as a solvent), and Mass Spectra (MS) were obtained with (agilent high resolution).
Synthesis
The new derivatives were synthesized in the successive reactions, as displayed in Scheme 1.

Synthesis of galloyl hydrazide (I)
Galloyl hydrazide was prepared following the MW-assisted procedure depicted by A.M. Rabie via a new green chemistry from gallic acid: off-white solid, mp 294-297 °C [11].
Synthesis of (Z)-3,4,5-trihydroxy-N'-(1-(4-substitutedphenyl) ethylidene) benzohydrazide [II]a-e
A mixture of galloyl hydrazide (II) (1.84 g, 0.01 mol) and substituted aromatic ketone (0.01 mol) in ethanol (10 mL) and four drops from glacial acid (GAA) was heated for (9 hours), and then cooled and the precipitate was collected by filtration, recrystallized from ethanol [12].
Synthesis of 3-substituted phenyl-5-(3,4,5-trihydroxyphenyl)-4H-pyrazole-4-carbaldehyde [III]a-e
Synthesis of compounds [III]a-e using POCl3 (0.01 mol) dripping to an ice-stirred solution of compound [II]a-e (1 mol) in dry dimethylformamide (10 mL), and the mixture was allowed to cooled, and then refluxed at 70 °C for (4 hours). Using a water bath, it was poured onto ice water, neutralized with dilute sodium hydroxide, and left standing (24 hours), recrystallization was by ethyl acetate [1]. The physical data of hydrazones and pyrazole derivatives [III]a-e are presented in Table 1.
Results and Discussion
Chemistry
The new hydrazones [II]a-e were produced by the condensation reaction of equimolar amounts from galloyl hydrazide I with different substituted aromatic ketones in ethanol and GAA. These compounds [II]a-e were investigated by FT-IR and 1H-NMR spectroscopy. The FT-IR spectrum of these соmроunds [II]a-e hаs shown the new stretch peaks оf С=N, and the disаррeаrаnсe оf the C=O grоuр and NH2 group together [13]. The FT-IR absorption stretching vibration peaks absorption of compounds [II]a-e are listed in Table 2. Likewise, the 1H-NMR-spectrum of compound [II]e exhibited the singlet type signal at δ = 9.29-8.98 ppm assigned to an OH for 3,4,5-trihydroxyphenyl moiety, also doublet of signals between δ 8.11-8.39 ppm which is due to four protons of aromatic rings and singlet signal at δ 8.08 ppm due to NH and a singlet signal at δ 6.95 ppm for the 2H aromatic ring. Furthermore, the singlet type signal at δ 3.76 ppm was assigned to three protons of the methoxy group. Therefore, the singlet type signal at δ 2.42 ppm was attributed to 3H for methyl moiety [13-15].


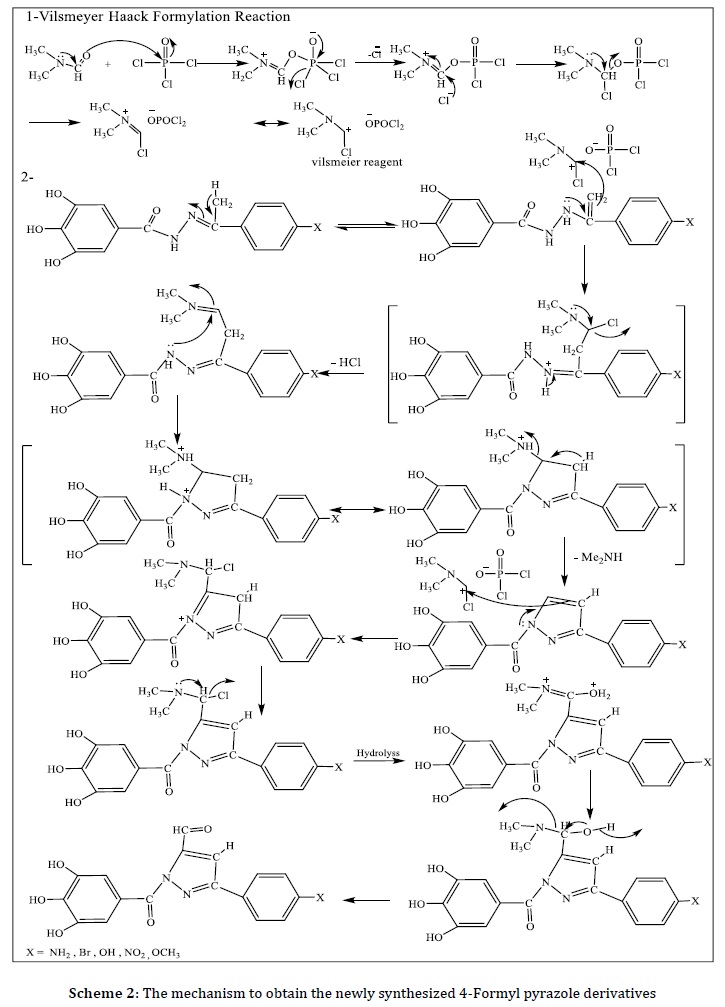
The 4-formyl pyrazole derivatives [III]a-e are produced via cyclization reaction of hydrazones [II]a-e mixing with POCl3/DMF, following Vilsmeyr Hatchh formylation reaction, as indicated in Scheme 2. The new 4-Formyl pyrazole derivatives [III]a-e were investigated by FT-IR, 1H-NMR, and mass spectroscopy. The FT-IR spectrum of for these compound, appearance of new stretching bands for C=O at position C-4 from pyrazoline ring, the FT-IR bands of aldehydic compounds [III]a-e were listed in Table 2. The 1H-NMR - spectrum of [III]b exhibited the singlet signal at δ 10.14 ppm could be attributed to the C-H aldehyde group, and the singlet signal at δ 9.91-9.71 ppm due to the O-H, also the multiplet signal at δ 6.94-7.58 ppm which is due to six protons of the aromatic ring and protone of pyrazole ring. The 1H-NMR-spectrum of [III]e displayed the singlet type signal at δ 9.29 ppm assigned to one proton of C-H aldehyde moiety and the singlet signal at δ 8.98-8.97 ppm could be attributed to the O-H, also the multiplet signal at δ 7.90-7.40 ppm for p-substituted benzene ring and proton of pyrazole ring, also at δ 6.91 ppm singlet signal for 2H-aromatic (3,4,5-trihydroxy benzene ring) and signal at δ 3.76 ppm is due to OCH3 moiety [16-20].
The mass spectrum of [III]a: Chemical Formula: C17H13N3O5, (M.Wt.=339.08) as depicted in Scheme 3. Figure 1 displayed the base peak at (m/z=105). Likewise, several fragments at m/z= 311, 294, 165, 126, 91, 77, and 65. The spectrum also showed peak at m/z=67 refer to the pyrazole ring.
Biological evaluation
The anti-microbial activity of the derivatives [III]a-e were examined using two kinds of bacteria (Staphylococcus aureus) (G+) and (Klebsiella pnumonia) (G-) in Muller Hinton Agar medium using Agar Well Diffusion Method [15, 21]. All the synthesized compounds [III]a-e were placed serially in the cavities with the help of a micropipette and allowed to diffuse for (1 hour). Dimethyl sulfoxide (DMSO) was used as a solvent for all the compounds at a concentration of (100 µg/mL). These plates were incubated at 37 °C for 24 hours, as compared with the common antibiotic Ampicillin. The zones of inhibition formed were measured in millimeters. The derivatives were evaluated and exhibited excellent to a good range of antibacterial activity against both selected bacteria. Table 3 presents the results of studies on both bacteria. The experimental performs detected that compounds [III]C and [III]e displayed promising antibacterial activity compared with the well-known antibiotic ampicillin.
Conclusion
Here we recorded an efficient project, in which the design of 3-aryl substituted pyrazole-4-carbaldehyde derivatives, and then synthesized these derivatives using method of Vilsmeier–Haack reaction. The galloyl hydrazide was used as starting materials to synthesizide the newly substituted 4-formyl pyrazole IIIa-e via cyclization process of hydrazone in POCl3/DMF.The structure illustration for these derivatives was subjected to spectral analysis by FT-IR, 1H-NMR, and mass spectroscopy. Given that a good inhibitory activity of the 4-formyl pyrazole based to galoyll hydrazide on both bacteria was identified with the purpose of investigating the inhibitory potency antibacterial activity.
Acknowledgements
The research is part of the PhD requirements for the researcher Fiadh A. Neshan.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
Conflict of Interest
We have no conflicts of interest to disclose.
ORCID
Fayyadh Abed Nashaan
https://orcid.org/0000-0002-7218-0675
Muna Sameer Al-Rawi
https://orcid.org/0000-0002-0318-8999
HOW TO CITE THIS ARTICLE
Fayyadh Abed Nashaan, Muna Sameer Al-Rawi. Synthesis and Antimicrobial Activity of New 4- Fromyl Pyrazole Derivatives Drived from Galloyl Hydrazide. Chem. Methodol., 2023, 7(4) 266-275


