Document Type : Original Article
Authors
Department of Chemistry, College of Education of Pure Science, Ibn – Al Haitham, University of Baghdad, Iraq
Abstract
A new furfural Schiff base derivative ligand (L-FSB) named N-(4-Bromo-2-methylphenyl)-1-(furan-2-yl)methanimine, was synthesized from the condensation reaction of furfural (fur) with 4-Bromo-2-methylaniline (bma) in 1:1molar ratio. A new series of VO(II), Cr(III), Mn(II), Co(II), Ni(II), Cu(II), Zn(II), and Cd(II) metal complexes are synthesized according to the metal content analysis in an 2:1 ligand:metal ratio. The stereochemistry of the ligand complexes have been deduced by Fourier Transform-Infra Red (FT-IR), Atomic Adsorption (A.A), Ultra violate-Visible Spectra (UV-Vis Spectra), (Mass Spectra, Proton,13Carbon-Nuclear Magnetic Resonance) (1H-NMR,13C-NMR) for ligand), magnetic susceptibility at 25oC and conductivity measurements. From the FT-IR data the coordination behavior of the ligand is a neutral monodentate ligand coordinate to the metal ions through azometane nitrogen. Electronic spectra analysis study revealed that the suggested geometrical structure of all complexes are distorted octahedral geometry except Zn(II) and Cd(II) complexes are tetrahedral and this study was confirmed by magnetic moment and conductance measurements. The antimicrobial activities of the synthesized Schiff base ligand and its complexes were screened against three pathogenic microbes: Escherichia coli (Gram-negative bacteria), Staphylococcus aureus (Gram-positive bacteria), and fungal strains (Candida albicans) were identified the most efficient biologically active compounds. The results of studies showed that potential is related to the structure and nature of ligand and their complexes.
Graphical Abstract
Keywords
Main Subjects
Introduction
The processes of designing drugs from organic compounds with new and practically important biological behaviors is required organic interactions involving the condensation of two or more types of molecules to obtain a new derived from imine compounds as a commonly used compounds for this purpose [1-4]. The reaction of condensation of the primary amines with aldehydes or ketones gives an important product. The Schiff base reaction was done under normal conditions. The presence of a Schiff base in our interactions is of great importance due to the stability of complexes of metal ions with different oxidation states and because of its involvement in many vital processes on a large scale such as industrial fields and as catalysts for reactions in addition to its significant wide involvement in biological activities [5]. The stable structure of metal complexes is attributed to the nitrogen lone pair electrons on azomethine (-N=CH) bonding [6]. Donor atoms of Schiff base ligand can be enhanced the antibacterial activity through the coordination to metal ions [7-9]. The interaction of metal ions with Schiff base ligands can give structures of different geometric shapes and have various applications such as their use in organometallic chemistry as anti-tumor, anti-cancer, anti-bacterial, anti-viral, and medicinal compounds designed, field of geology, as catalysts, anti-bacterial, anti-fungal agents, antiviral agents and for the design of important medicinal compounds [10, 11]. The effect of PH and concentrations on donor tetradentate ligands type N2O2 on the formation of transition metal complexes have been studied [12]. New Schiff base mixed ligand complexes of β-Enaminone with 8-hydroxyquinoline and Oxalic Acid with Schiff base trimethoprim chelates play an important role in biological processes such as the activation of transport substances across membranes, storage, and enzymes [13, 14]. Amines derived from furfuralsor furfurylamines present diverse applications such as industry, pharmaceutical compounds, synthetic resins, polymers, agrochemicals, antiseptic agents, and pesticides as well as Barmastine and an anti-hepatitis-B [15, 16]. In this paper, we have been synthesized, characterized, and conducted antimicrobial studies of new Schiff base ligand N-(4-Bromo-2-methylphenyl)-1-(furan-2-yl)methanimine (L-FSB) and its metal complexes with: VO(II), Cr(III), Mn(II), Co(II), Ni(II), Cu(II), Zn(II), and Cd(II).
Materials and Methods
All used chemicals reagents were of the analytical propose and the highest purity from (BDH). All the organic solvents are absolute, Dimethysulphoxide (DMSO) ethanol and diethylether were either spectroscopic pure from (Aldrich).
Instruments
The melting point was registered on “G. Kamp, device model SMP30 at College of Education of Pure Science, Ibn Al-Hathiam. FT-IR are performed in the range of 4000-400 cm-1 in KBr discs and in the range of 4000-200 cm-1 in CsI discs on (FT-IR) Shimadzu type-8400S. Spectra were recorded at Laboratories of the College of pure Sciences, University of Diyala. 1H-NMR and 13C-NMR of the new ligand was recorded by using (Brucker-500 MHz spectrophotometer) was measured in DMSO-d6. The chemical shifts are in ppm relative to TMS were performed at University of Tehran, Iran. GC–M spectrum was performed using Agilent mass spectrophotometer, 5973N.S.D Mass, at the laboratory of University of Tehran, Iran. Magnetic susceptibility values were obtained at 25 ᵒC using Auto Magnetic susceptibility Balance Sherwood Scientific using the Faradays method at Al-Mustansiriya University. TLC for the ligand (L-FSB) was performed in aluminum plates coated by (0.3 mm) layer of silica gel Fluka type (F254) and detected in Iodine. Conductivity measurements were registered using conductivity meter type VIRA, B3000 in DMSO solvent at laboratory of (BPC), Baghdad. UV-Vis spectroscopy were measured by using (SHIMADZU-UV-240) at the wavelength range (200–1100 nm), using match quartz cells (1 cm) and DMSO as a solvent at laboratory of (BPC), Baghdad. The metal content of complexes were measured using Shimadzu Emission spectrophotometer type AA (680) at Ibn Sina Company, Ministry of Industry Baghdad, Iraq.
Biological activities
Method of preparation of agricultural media
The nutrient media (Muller Hinton agar) prepared from (mast group) was prepared by gram of powder in a liter of distilled water, and (pH = 7.3), and then sterilized by Auto Cleave. This medium was used for the purpose of growing microorganisms and studying the antagonistic activity of chemicals against the isolates used of this study.
Preparation of microorganism suspension
The suspension of microorganisms was prepared by taking colonies from each of the microorganisms under study, and placing it in sterile physiological saline at a concentration of 0.85%. The Petridis dishes were used in its preparation to examine the biological activity against the chemical compounds under study.
Microorganisms testing the inhibitory activity of complexes in facultative
The agar well diffusion method was used to observe the effect of ligands and chemical complexes under study on the growth of microorganisms by pouring 20-25 millimeter of agar medium into each plate and keeping the plate after it was cooled in the incubator for a whole day (1 hour) at 37 ᵒC to ensure that there is no contamination of the dishes. The plate was soaked with 10 mL of the prepared microorganism suspension as mentioned in the previous paragraph containing 1.5×810 cells/mm, and then spread evenly on the surface of agar medium using a glass spreader, a hole was made on the surface of the culture medium with a glass spreader. Chemical complexes by 100 micro ml for each well at a concentration of 1×10-3 M, with the remainder of control hole containing the solvent (DMSO), the dishes were left for half an hour in the refrigerator at 4 ᵒC, and after that they were incubated in the incubator at 37 ᵒC .
Synthesis of ligand N-(4-Bromo-2-methylphenyl)-1-(furan-2-yl)methanimine (L-FSB)
2.58 g, (26.8 mmol) of furfural was taken in a conical flask with a capacity of 100 mL, and then 15 mL of absolute ethanol was added to it, the mixture was left to stirring for 10 minutes. After that three drops of glacial acetic acid were added to the mixture, and the stirring continued for 10 minutes (5 g, 26.8 mmol) of (2-amino-5-bromotoluene) dissolved in absolute ethanol (25 ml) was added to it. The mixture was refluxing and stirring at 70-80 ᵒC for 5 hours, while the reaction was monitored through TLC technique (Rf = 0.84) , the solution was cooled, and then the precipitate was filtered and left to dry giving a pale yellow precipitate, and recrystallized using hot ethanol.
Synthesis of (L-FSB) metal complexes
Synthesis of [Cr (L-FSB)2 (H2O)2 Cl2 ] Cl . 8H2O complex
(0.15 g, 0.56 mmol) of chromium trichloride hexahydrate (CrCl3.6H2O) was dissolved in 5 mL of absolute ethanol, and then added gradually to the mixture (0.2 g, 1.21 mmol) of ((L-FSB) ligand dissolved in 20 mL of absolute ethanol). The mixture was left for continuous stirring for half an hour, and then the mixture was refluxing at a temperature 70-80 ᵒC for 3 hours. Thereafter, the solution was left to dry at room temperature forming a violet precipitate, and then the precipitate was collected and recrystallized with hot ethanol, to obtain dark violate precipitate .
Synthesis of [(M)n(L-FSB)n (Y)n (X)n] nZ .nb complexes
[(L-FSB) = Furfural Schiff Base Derivative Ligand]
[Y = H2O, X = Cl] Z = (SO4) for VO(II) complex only or Cl, b = H2O,
n = (1 , 2 , 3 , 4, ...)
Mn (II) = (VO)2 , Mn, Co, Ni, Cu, Zn and Cd
(L-FSB)n = 4, 2, 2, 2, 2, 2 and 2
(Y)n = 4, 2, 3, 3, 3, 1 and 0
(X)n = 0, 2, 1, 1, 1, 1 and 2
(Z)n = 2, 0, 1, 1, 1, 1 and 0
(b)n = 18, 7, 2, 5, 3, 4 and 11
The same previous method was followed in paragraph (2-5-1) to synthesize the following metal complexes using of (1 mmol) of metal ion salts: (0.180 g, VOSO4.H2O), (0.197.93 g, MnCl2.4H2O), (0.237 g, CoCl2.6H2O), (0.237 g, NiCl2.6H2O) , (0.170 g, CuCl2.2H2O), (0.136 g, ZnCl2) and (0.201 g, CdCl2.H2O) with (2 mmol) of the ligand (0.528 g, (L-FSB)) to give different colors of complexes precipitates which were collected by filtration, washed with ethanol, and then vacuum dried.
Results and Discussion
Synthesis and physical properties of (L-FSB)`ligand and It's metal complexes
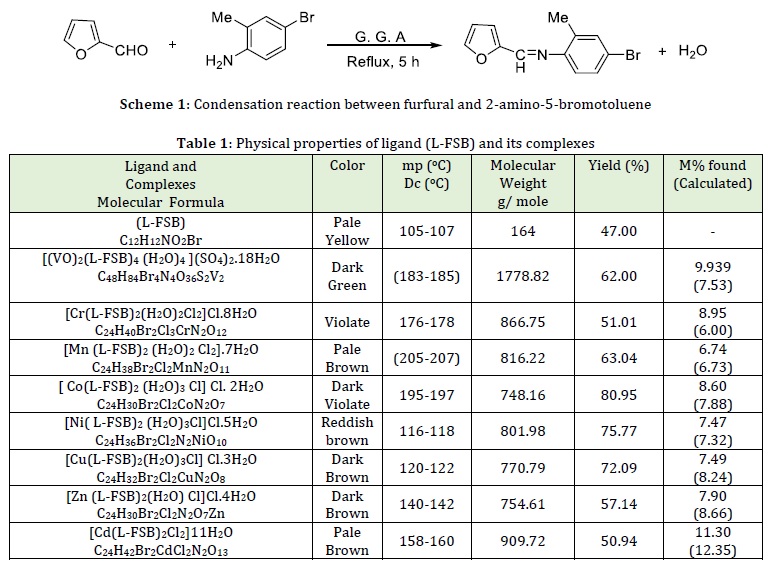
The new Schiff base (L-FSB) was prepared as a furfural Schiff base derivative by the condensation reaction of furfural with of 2-amino-5-bromotoluene in a mole ratio (1:1), respectively. The reaction is illustrated in Scheme 1.
The formation of (L-FSB) ligand was characterized and confirmed by melting point, TLC, FT-IR, UV-Vis, 1H-NMR, 13C-NMR, and mass spectra data. It was found to be stable towards air. This ligand was sparingly soluble in methanol ethanol, acetone, chloroform, ethyl acetate, benzene, and soluble in DMSO, DMF, and THF diethyl ether, but insoluble in water. The coordinating behavior of synthesized (L-FSB) ligand with their metal ions (VO(II), Cr(III), Mn(II), Co(II), Ni(II), Cu(II), Zn(II), and Cd(II) have been studied. Analytical data of metal content for the ligand (L-FSB) and its metal complexes suggest that (metal: ligand) was (1:2) stoichiometry. Most of complexes have high melting points and show stability. The physical properties of ligand (L-FSB) and their complexes are presented in Table 1.
Spectroscopy studies
FT-IR spectra of the ligand (L-FSB)
The FT-IR spectrum of the new ligand (L-FSB) appeared of a sharp, split, and a new band at 1630 and 1624 cm-1, which is due to the formation of azomethine. υ (CH=N) is an indication of the condensation of amino group NH2 of 4-Bromo-2-methyl aniline compound with the aldehyde group of the furfural and its converted to azomethine group [17].
FT-IR spectra of ligand (L-FSB) and its metal complexes
The infrared spectrum of the free Ligand (L-FSB), Figure 1 appearing band at 1631 and 1624 cm-1 attributed to azomethine was shifted to lower frequency by 4-11 cm-1 in all complexes except copper complex was shifted to higher frequency about 62 cm-1 with change in the intensity of band of cadmium complex indicating to the participation of nitrogen atom of azomethine in the coordination of complexes [18]. The ligand (L-FSB) showed broad band at 3200 cm-1 due to presence of water molecular. In metal complexes showed broad and strong absorption bands in the range of 3680-3221 cm-1 due to the OH stretching vibration of lattice water [19] while the presence of coordinated υ(M-O) was detected by the appearance of bands at the range of 594-509 cm-1 in all complexes [20] except for Cd(II) complex. Another evidence of presence of the coordinated bands of aqua water υ (M-OH2) were appeared of new bands at in the range 867-987 cm-1 in all complexes except Cd(II) complex. Another new bands was appeared in VO(II) complex, Figure 2 at 979 cm-1 and 1041 cm-1 which is due to stretching vibration of υ (V=O) and υ (SO4)-2 free ion [21]. On the other hand all complexes showed new bands at the range of 432-478 cm-1, due to υ (M-N) and another new band at the rang of 275-378 cm-1 except for VO(II) complex, is due to the stretching vibration of υ(M-Cl), and that confirmed the coordinated behavior of (L-FSB) was mono dentate through the coordinate by nitrogen atom of azomethine group [22]. All prominent absorption bands of the ligand (L-FSB) and its metal complexes are presented in Table 2.


1H-NMR of the ligand (L-FSB)
The 1H-NMR spectra of the ligand (L-FSB), Figure 3 appeared one signal at δ = 2.31 ppm due to 3, protons of methyl group [23], and multiplet signals at the range of δ = 6.99-7.39 ppm due to 3 protons of aromatic ring, another signals at the range of δ = 6.65-7.40, which due to the 3 protons of furan ring. The singlet signal at δ = 8.31 ppm due to the H proton of the azomethine group and finally the chemical shift at δ = 2.52 ppm is due to the DMSO solvent [24].
13C-NMR of the ligand (L-FSB)
The spectrum analysis of 13C-NMR for the prepared ligand are present in Figure 4 using DMSO-d6, shows the signal at δ = 17.54 ppm were attributed to the carbon atom of methyl group, and multiple signals at the positions δ = 121.15, 123.98, 129.75, 132.42, and 134.8 ppm due to the carbon atoms of the aromatic ring and also the spectrum shows another multiple signals at the positions δ = 112.36, 118.46, 147.08, and 150.03 ppm which due to the carbon atoms of furan ring. The signal position for carbon atom of azomethine group was appeared at δ = 145.53 ppm [25].
Mass spectra fragmentation of the ligand (L-FSB)
The mass spectra of the furfural Schiff base ligand exhibit parent ion peak due to the respective molecular ion [M] and that is corresponding to the molecular weight of free ligand (L-FSB) and indicated to its purity. The molecular ion peak of the ligand C12H11NOBr appears at m/z = 264, which is in good agreement with the theoretical value. The other fragments peaks are detected at m/z = 250, 235, 184, 183.2, and 171 corresponding to ions fragments. All these fragments and their abundance can be shown in Table 3 and Figure 5 [26, 27].
The UV-Vis spectrum of the pale yellow N-(4-Bromo-2-methylphenyl)-1-(furan-2-yl)methanimine (L-FSB) in DMSO, Figure 6 solvent exhibited two absorption bands and high intensity at 288 nm and 34222 cm-1 and 330 nm and 30303 cm-1 are assigned to π→π* and n→π* for electronic transition of aromatic or furan ring and azomethine group, respectively [28].
The UV-Vis spectrum of the dark green oxovanadium(II) complex
All the complexes displayed higher intensity absorption bands, in the UV region which attributed to ligand field or charge transfer band. The other two bands have lower intensity appearing at 380 nm, 26316 cm-1, and 836 nm, and 11962 cm-1 attributed to (d-d) electronic transition type 1B2→ 1A1 and 1B2 → 1E electronic transition, respectively. According to these results and in addition to the results of FT-IR, metal content, and confirmed with the results of (Xg =0, Dimeric) as well as magnetic moments at room temperature and molar conductivity in DMSO solvent was 1:1 electrolyte, we can suggest the di-nuclear square pyramidal geometry of (VO)(II) complex [29] (Figure 7).




UV-Vis spectrum of violate chromium(III) complex shows a band with broad intensity at 502 nm and 19220 cm-1 is attributed to (d-d) electronic transition type 4A2g→ 4T1g(P) υ3 and the other (d-d) transition bands did not appears or is very weak, based on these results of magnetic moments of Cr(III) was (3.85 B.M ) at room temperature and the molar conductivity value in DMSO was 1:1 electrolyte in addition of these results, FT-IR, and metal content, we suggested the distorted octahedral geometry of Cr(III) complex [30].
UV-Vis spectrum of pale brown manganese(II) complex appeared an absorption band at 352 nm and 28409 cm-1 is attributed to the charge transfer band. The measurement of magnetic susceptibility of manganese(II) solid complex showed a value at 5.49 BM at room temperature, which is due to the presence of five individual electrons with high spin state, and this supports by the measurements value of molar conductivity in DMSO solvent was non-electrolyte in addition of these results, FT-IR, and metal content, we suggested the distorted octahedral geometry of Mn(II) complex [31].
UV-Vis spectrum of dark violate Cobalt(II) complex exhibiting two absorption bands at 602 nm and 16111 cm-1 and 672 nm and 14881 cm-1, these bands are observed in the visible region center at 645 nm and 15496 cm-1, this may be attributed to the transition 4T1g (F) → 4T1g(F) υ2), the other, electronic transition bands type 4T1g(F) → 4T2g(F) υ1 and 4T1g → 4T1g(P) υ3 could not detected or is very weak , so besides studying magnetic moments at room temperature of Co(II) complex was (2.3 B.M) confirmed high spin of d7 ion and that indicate the paramagnetic nature. The measurement value of molar conductivity in DMSO was 1:1 electrolyte. According to these results in addition to the results of FF-IR and metal content, we suggested the distorted octahedral geometry of Co(II) complex [32].
UV-Vis spectrum of reddish brown Nickel(II) complex show electronic transition band with high intensity at 404 nm and 24752 cm-1 is attributed to (d-d) electronic transition type 3A2g→ 3T1g(P) and the other two electronic transition bands type 3A2g → 3T1g(F) and 3A1g → 3T2g(F) could not detected or is very weak. The measurement of magnetic moment of nickel(II) solid complex showed a value at 2.86 B.M at room temperature, which is due to the presence of two individual electrons with high spin state, and this supports by the measurements value of molar conductivity in DMSO solvent was (1:1) electrolyte in addition of these results, FT-IR, and metal content, we suggested the distorted octahedral geometry of Ni(II) complex [33].
UV-Vis spectrum of dark brown Copper(II) complex exhibiting weak intensity adsorption band at 882 nm, 11338 cm-1 can be assigned to the electronic transition type (2B1g → 2B2g). On the basis of these results, the measurements of magnetic moment value of the solid Cu(II) is 2.34 B.M. at room temperature and also confirming with the results of molar conductivity in DMSO solvent is (1:1) electrolyte, in addition to the results of FT-IR of metal content, we suggested the distorted octahedral geometry of Cu(II) complex [34, 35].
UV-Vis spectrum of dark brown Zinc(II) complex and pale brown Cadmium(II) complex. The existence of tail that which extends to the range of 420 nm, is due to the charge transfer spectrum of Zn(II) and Cd(II) complexes. The Zn(II) and Cd(II) complexes are diamagnetic, and the molar conductivity of Zn(II) complex is (1:1) electrolyte and for Cd(II) complex is non-electrolyte, so according to these transitions and in addition to the results of FT-IR and metal content, we suggested the tetrahedral geometry of Zn(II) and Cd(II) complex [36]. The UV-Vis Spectra data, magnetic moment, and molar conductance results of (L-FSB) complexes are listed in Table 4.
Antimicrobial activity studies
The antimicrobial studies of the newly synthesized furfural Schiff base ligand (L-FSB) and their metal complexes have tested against three pathogenic microbes as Gram-positive (Staphylococcus aureus), (Escherichia coli), and fungal strains (Candida albicans) using the disk diffusion method. The test (L-FSB) ligand and metal complexes were dissolved in (DMSO) to get a solution of 10 mg/mL [37, 38]. The biological activity studies of (L-FSB) and complexes are presented in Tables 5 and Figure 8.
Biological activity of prepared ligand (L-FSB) and its complexes against bacterial and fungal. The prepared ligand (L-FSB) showed inhibitory activity with the same amount against positive and negative bacteria. All its derived complexes showed the activity against Gram-negative and Gram-positive bacteria in various proportions. Cadmium(II) and Cobalt(II) complex is more effective than other complexes against bacteria (Staphylococcus aureus), while the complex of Cadmium(II) and Nickel(II) showed higher inhibitory activity than the rest of the complexes against the bacteria E.coli.
With regard to fungus, the prepared ligand (L-FSB) showed high inhibitory activity against Candida albicans compared with its activity against bacteria and also all the complexes derived from it showed effectiveness and in varying proportions, copper(II) and cadmium(II) complex showed a higher inhibitory activity than the rest of the complexes prepared against fungus. The reason for the effectiveness of the prepared ligand and its complexes against staph bacteria and E.coli bacteria are due to several reasons, including:
1- The solutions of these ligands and their complexes have the ability to dissolve the fatty layer of wall of these bacteria, which causes the liquids in the cell to leak out and destroy them.
2- The chelating nature of the prepared ligand (L-FSB), as it can form complexes with the metallic elements present in the cell body such as magnesium, zinc, calcium, copper, cobalt, and iron that microorganism’s need, which strips these bacteria of these essential elements and disables them from her death [39, 40].


Conclusion
A new series of metal complexes of VO(II), Cr(III), Mn(II), Co(II), Ni(II), Cu(II), Zn(II), and Cd(II) have been coordinated with new Schiff base derived from (bma) and (fur), in an (1 to 2) ratio (metal to ligand) based on the results of metal content (A.A). All of these compounds characterized using different techniques: FT-IR, UV-Vis spectra, (1H-NMR, 13C-NMR, and mass spectra for ligand), magnetic moment and molar conductivity. FT-IR explains the metal coordinated through nitrogen atom of azomethine and oxygen atom of water. The FT-IR results of the ligand (L-FSB) were in good agreement with those obtained from 1H-NMR, 13C-NMR, and mass spectra. Conductance measurements values showed that VO(II), Cr(III), Co(II), Ni(II), Cu(II), and Zn(II) complexes are 1:1 electrolyte except Mn(II) and Cd(II) are non-electrolyte. The study of UV-Vis spectra and magnetic moment measurements support and confirmed the geometrical structure for all complexes are distorted octahedral except Zn(II) and Cd(II) complexes are tetrahedral. The biological analysis of (L-FSB) and its complexes against Staphylococcus aureus (G+), E.Coli (G-) bacteria, and Candida albicans was tested to understand the effect of binding metal ions to the (L-FSB) ligand. Antimicrobial study indicates that metal complexes show more inhibition than (L-FSB) ligand.
Acknowledgments
The authors would like to gratefully thanks the Ministry of High Education and scientific research to give this opportunity to complete this research, especially thank to the emeritus prof. Dr. Falih Hassan Musa, for his evaluation of this research and for his scientific information, which had an important role in completing the work . Finally, I extend my sincere thanks, to all who contributed to provide scientific assistance and advice.
Disclosure Statement
No potential conflict of interest was reported by the authors.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' Contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
Orcid
Noor S. Hassan
https://orcid.org/0000-0001-6407-6926
Waleed K. Mahdi
https://orcid.org/0000-0002-4097-3945
HOW TO CITE THIS ARTICLE
Noor S. Hassan, Waleed. K. Mahdi. Spectroscopic and Antimicrobial Studies of Some Metal Complexes of Furfural Schiff Base Derivative Ligand. Chem. Methodol., 2023, 7(6) 419-434


