Document Type : Original Article
Authors
Department of Chemistry, College of Science for Women, University of Baghdad, Iraq
Abstract
To reflect modern practice in lab experiments, they should include a range of elements. Mixed new complexes of metal chloride for Rh(III) and Au(III) synthesis by the condensation reaction of metals with sodium fusidate as a primary ligand with the secondary ligand represented by 2,2-bipyridine (L/) in boiling ethanol and synthetic tasks are accompanied by characterization and analysis in the solid state based on the results of the color, melting points, metal composition percent and other elements (C, H, and N), the spectroscopic measurement acts by UV-Vis and FT-IR, magnetic susceptibility, and electrical conductivity measurements. The results of this study reveal the coordination sites for the ligands with the metal ions as bidentate through two oxygen atoms of the sodium fusidate, while bipyridine via diimine nitrogen atoms. Analytical and spectroscopy studies disclosed octahedral structure for Rh(III) complex and square planner structure of Au(III) complex. The ligands and both inclusions of new complexes were screened against bacterial and fungal strains (Escherichia coli as gram (-), Staphylococcus aurous as gram (+) and Candida albicaons) at three selected concentrations (50, 100, and 200 ppm) and their preliminary results compounds exert the cell surface damage observed by scanning electron microscopy (SEM) and the Au(III) complexes exhibit the superior antimicrobial activity particular in a high concentration (200 ppm).
Graphical Abstract
Keywords
Main Subjects
Introduction
The most recent studies have improved the study of transition metal ions in coordination chemistry with various types of ligands in the realms of medicine and bioinorganic chemistry [1]. Sometimes, metal complexes of biologically significant ligands are more efficient than the ligands alone [2]. The stability of various oxidation states, solvophilicity, and the electrical characteristics of the metal ions are all governed by the type of ligands used in complexes [3]. As millions of possible ligands are expected to compete for metal ions in the human body, the mixed ligand complexes play a crucial role in biological chemistry because mixed chelation occurs frequently in biological fluids [4]. These create specific structures and have been implicated in the storage and transport of active substances through membranes [5]. The latter characteristics are crucial to the therapeutic effects of transition metal coordination compounds. Thus, pharmaceutical corporations have considered strategies to avoid the spread of commercially available drug-resistant microorganisms [6, 7]. Fusidic acid (FA) is a naturally occurring, effective antibiotic that is made by the fungus Fusidium coccineum. It is a thermostable lipophilic tetracyclic triterpenoid [8]. The isolation process was initially documented by Godtfredsen et al. [9, 10] of LEO company pharmaceutical products, Denmark, in the early 1960s. Initially, the fungus had to be grown for 120 hours at 24 °C in deep culture fermenters before the antibiotic could be isolated directly from the growing media after being clarified [9, 10]. According to its utilization, it is mostly utilized in Europe and Asia to treat methicillin-resistant Staphylococcus aureus infections. Although many derivatives of fusidic acid have been created, sodium fusidate was the only one that was known [11]. 2,2-bipyridine ligand is an excellent chelating bidentate ligand. Its σ-donation is accomplished by the π-acceptor ability enhancing the complex stability [12, 13]. This enables metal complexes to have a clearly defined spatial configuration [14], which results in increased antibacterial activity [15].
Therefore, in this study, new Rh(III) and Au(III) complexes and precursor ligands are investigated for their structural properties and biological activity. The bioactivities of the uncoordinated ligands and metal ions are found to be combined in mixed-ligand complexes, giving them the potential to be more effective antimicrobial agents.
Materials and Methods
Sodium fusidate in a purity 99.5%, RhCl3.H2O, and HAuCl4.H2O (analytical grade), and the solvents used in the work were supplied from Merck (Schnelldorf, Germany). Stuart Melting Point Apparatus was used to measure the melting points of the obtained complexes. The UV-Visible spectrophotometer UV-1601 was employed to determine the absorbance wavelength by using a quartz cuvette and ethanol as the reference. The Fourier Transform Infrared (FT-IR) spectra were recorded for free ligands and metal complexes in the 4000-200 cm-1 on an FT-IR 8300 Shimadzu Spectrophotometer. Elemental microanalysis C.H.N was accomplished on the metal complexes that were nominated through the micro analytical unit at Euro EA3000 elemental analyzer. Atomic absorption was conducted by using (Atomic Absorption Spectrophotometry AA-6880 Shimadzu). The molar conductivities were measured for of complexes at 10-3 M in solution of ethanol complexes at 25 °C by using ProfiLine Oxi 3205 Instrument. The magnetic susceptibility measurements were obtained at R.T by using (MSB–MKI) magnetic susceptibility moder balance. All the steps of antimicrobial test were carried out at microbiology laboratory.
Synthesis of new metal complexes
A sodium fusidate (essentially ligand; L) dissolved in ethanol and 2,2-bipyridine (co-ligand; L), about 10 mL of mixture solution of the ligand, were added slowly to the metal salts of RhCl3.H2O and HAuCl4.H2O dissolved in ethanol by using stoichiometric amounts [(1:1:1) (M:L:L)]. The mixture was heated and reflux for 2 hours. The color crystalline precipitates were observed, dried in a vacuum, and recrystallized from ethanol after being filtered and repeatedly irrigated with water to remove NaCl (Scheme 1). All the prepared complexes were identified by using spectroscopic, analytical, and physical measurements, as represented in Table 1.
Determination method of antimicrobial activity
Sodium fusidate L and coligand bipyridine beside their heavy metal complexes of Rh(III) and Au(III) were evaluated in vitro by using Kirby–Bauer test method [16] against the development of two harmful bacterial isolates, one Gram-positive staphylococcus aurous and one Gram-negative Escherichia coli, as well as anti-fungi against candida albicaons. The antimicrobial activity was carried out on nutrient agar plates that had previously been injected with 24 hours-old broth cultures of the bacterial strains. The investigated compounds were put onto pre-sterilized filter paper discs (Whatman No. 3, 6 mm in diameter), which were then allowed to dry in a laminar flow biological safety cabinet. The discs were evenly spaced and positioned aseptically on the inoculated, hardened plates' surface. To extract diffusion and reduce the impact of time differences between the applications of various solutions, all plates that had been infected with bacteria and fungus were maintained in the refrigerated at 4 °C for 1 hour. A distinct zone around the discs, which signified the existence of suppression of bacterial and fungal growth, was found after the plates cultured for bacteria and fungus for 24 hours at 37 °C. The diameter of the inhibitory zones was measured (by using paper discs) and the data was gathered in millimeters.

Scheme 1: Mixed ligand complex formation reaction
Results and discussion
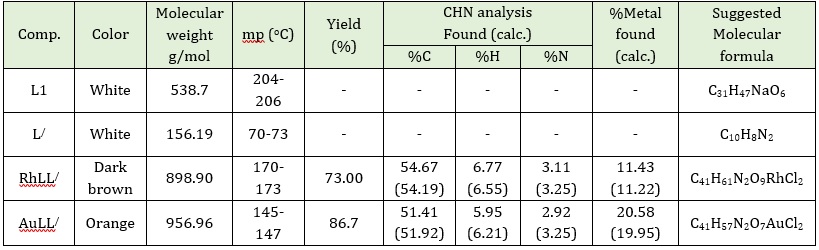
The ligands used in this work L and co-ligand L/ are white and the metal complexes are colored with their good percentage yields ranging from 73% and 86.7%, as indicated in Table 1. Both prepared compounds are steady toward light and air. The metal complexes were characterized by several analytical methods like C.H.N elemental analysis, A.A metal analysis, UV-Vis, FT-IR, magnetic moment measurements, and the molar conductivities. It was noted that the results of the analytical and experimental data are in agreement. The result proves that complexes ionic nature of both complexes and the value of magnetic susceptibility of the prepared complexes is assent with proposed molecular geometry of them. Physiochemical characters of ligands and their metal complexes are listed in Table 1.
Table 1: Physiochemical information of the primary and secondary ligands as well as two metal complexes
FT-IR spectra of ligands and their complexes
In the infrared spectrum of essential ligand (L), we can assign the valence vibration of the carboxylic group υC=O at 1716 cm-1 and other than of C-H, δ C-C appeared at 2947 and 1444 cm-1, respectively [17, 18].
The spectrum of these prepared complexes showed the vanished υC=O and emergence of addition peaks at about 1534 and 1530 cm-1 and 1377 and 1379 cm-1, successively due to asymmetric and symmetric carboxylate stretching vibration for both of them. ∆υ=200 cm-1 indicated the acting of this ligand as a bidentate behavior of this carboxylic group and it was coordinated with metal atoms similar [17]. New weak bands were seen in the complexes' spectra at 532 and 559 cm-1, respectively, which attributed to the stretching frequency of the (M-O) bond.
The infrared spectrum of bipyridine illustrated the main bands which assign to the stretching vibration of C=N + C=C appeared at 1604-1481 cm-1 and 1509-1450 cm-1, respectively. All the bands shift towards a high wave number in the spectrum of both complexes, thus indicating the coordination to metals via the two sp2 hybridized nitrogen atoms [17, 19]. More evidence for this coordination was the new medium or weak bands appeared at 260 and 273 cm-1, respectively and this supported the attachment of (M-Npy).
Also, the broad bands were around 3490-3425 cm-1 and 3478-3410 cm-1 belonging to the presence of water molecules out of sphere, another one appeared at 983 and 880 cm–1 is due to rocking and wagging bendy of water molecules within the field of coordination in Rh(III) complex, as demonstrated as in Table 2 and Figures S1, S2, S3 and S4 (Supplementary data).
Table 2: Selected FT-IR absorption bands in (cm-1) of mixed ligand complexes

UV-Vis of ligands and their complexes
Three basic bands were seen in the electronic spectra of free ligand, the first of which was an absorption band that initially emerged at 44444 cm-1 owing to an inter-ligand transition π→π* that occurred on the pi-system. In a different group, the π→π* transition also produces the second absorption band, which is positioned at 41322 cm-1. The third absorption band, which formed at 37037 cm-1, is attributable to the site of the n→π* electronic transition on the oxygen atoms (carboxylate, ester, and OH) [20], as listed in Table 3. The co-ligand (L/) (2, 2′-Bipy) showed absorption bands at 42553 and 34722 cm-1 which have been assigned as π→π* and n→π* transitions due to C=C and –N=C- groups, respectively [21, 22]. Due to the metal-ligand interaction, these bands cause a shift in the produced complexes, clearly indicating the coordination of the ligand to the metal ion.
The UV-Vis spectrum of the prepared Rh(III)-complex in solution state displays a weak band at 12945.31cm-1 due to the forbidden transition 1A1g →3T1g. This might happen if the promoted electron spins differently, resulting in the two T termed 3T1g and 3T2g [23], the bands appeared at 12345 and 21505cm-1, respectively correspond to 1A1g→1T1g and 1A1g→1T2g transitions [24]. Another band appeared at 29761 cm-1 due to charge transfer L →RhCT. The μeff of this complex is record 0.00 B.M. as expected as a diamagnetic [25]. This value came with published octahedral geometry around Rh(III)-complex. Au(III)-complex spectrum exhibits two bands in the positions 26666 and 33670 cm-1 belong to the transitions 1A1g→ 1B1g and 1A1g→1Eg, respectivley [26], and the other peak appeared at 34482, 35460, and 47619 cm-1, respectively attributed to charge transfer in a square planar geometry. The another data conform the suggested geometry of Au(III) complex i.e. diamagnetic moment [27]. According to these data, in addition to the information obtained from infrared spectrum, atomic absorption and elemental analysis, a octahedral geometry and square planar structure can be postulated for Rh(III) and Au(III) complexes successively, as demonstrated in Figure 1. The conductivity measurements of these two complexes indicates that they are electrolytic and that chloride ions are present outside of the coordination sphere [2]. Table 3 and Figures 2, 3, 4 and 5 illustrate the electronic spectra of complexes and their ligands.
Table 3: Probable assignment of electronic spectra of two ligands and their metal complexes, conductivity measurements, magnetic moments, and proposed geometry of synthesis complexes
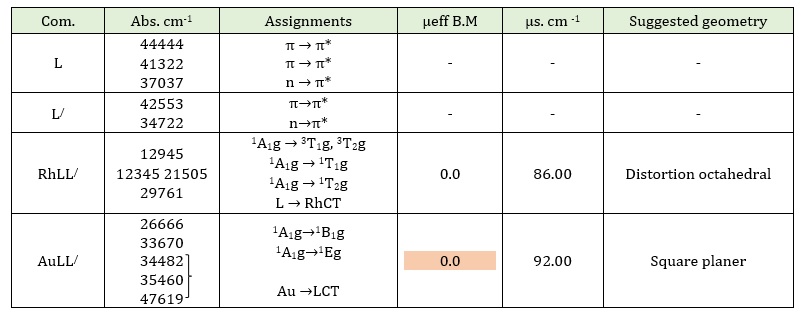
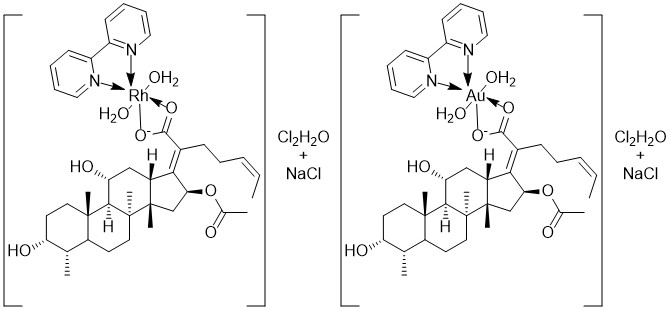
Figure 1: The proposed structures of new prepared complexes
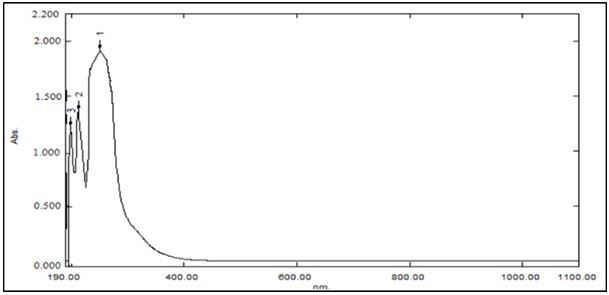
Figure 2: FA electronic spectrum
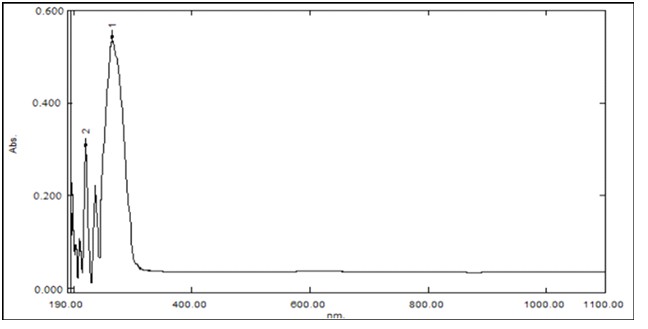
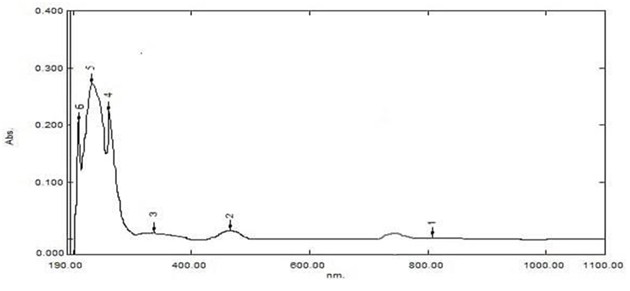
Figure 4: RhLL/ electronic spectrum

Figure 5: AuLL/ electronic spectrum
Antimicrobial activity
The antimicrobial action of the compounds, sodium fusidate, 2,2/-bipyridine, RhLL/(III), and AuLL/(III) was tested against two strains of bacteria consisting of one gram-negative, one gram-positive bacteria, and against fungal. The inhibition zone of the ligands and inclusion complexes are presented in Table 4. Testing for anti-biological activity against the microorganisms under the testing of ligands and their heavy metal complexes showed the presence of suppression zones of bacterial growth around the impregnated paper disks in the vast majority of the plates. The own metal complexes M(III) are more active compared with their parent ligands vs. the same microorganism and under the congruous trial conditions especially the gold complex with inhibitory zone range from 15.00 to 30.00 mm. Sodium fusidate, 2,2/-bipyridine zone diameter breakpoints was obtained after 24 hours of examination. It can be concluded that the sodium fusidate and its inclusion complexes exhibit a good antimicrobial activity especially in a high concentration. Binding to EF-G-GDP, sodium fusidate prevents the proteins synthesis by bacteria. This action also prevents peptide translocation and ribosome disassembly. There is limited cross-resistance with other known antibiotics since it has a new structure and manner of action [29].
Contrarily, 2,2'-bipyridine displayed the antibacterial activity because of its planarity and prolonged conjugation. It may intercalate with double strands of DNA to generate and contacts with base pairs preventing replication [30, 31]. The increased biological activity of these chelating complexes may be a result of the role that metal complex ions play in regular cell function. The complexes are thought to have soft metal ions, making them more lipophilic and facilitating their passage through the cell wall and influencing the cellular environment [32].
The better activities demonstrated by both ligands and inclusion new mixed complexes E. coli, S. aurous, and Candida albicaons can be described depending on overtone’s concept and Tweedy’s chelation theory [33]. The rhodium complex demonstrated a moderate activity compare with the gold complex which exhibits a great enhancement of activity against all micro-organism where chelation reduced the polarity of the metal ion by partially sharing its positive charge with the donor groups, which enhanced the delocalization of the pi-electron and increased lipophilicity, favoring absorption through the bacterial membrane to kill the cells [34].
This difference in synergetic effect between the type of metal ion and the two attached ligands may be attributed to many agents. The chelating affection of organic molecules used as ligands, central metal ion, and the atoms species that coordinated with metal ions, the oxidation state, and coordination number the arrangement of the ligand around the central ions and the final geometric shape of theses complexes [35, 36]. Finally, the data included in Table 4 and Figure 6 refer to both tested gram (+) and gram (-) bacterial strains and fungal were sensitive to free ligands and new prepared complexes.
Table 4: Inhibition zone of free ligands and their metal(III) complexes in ppm
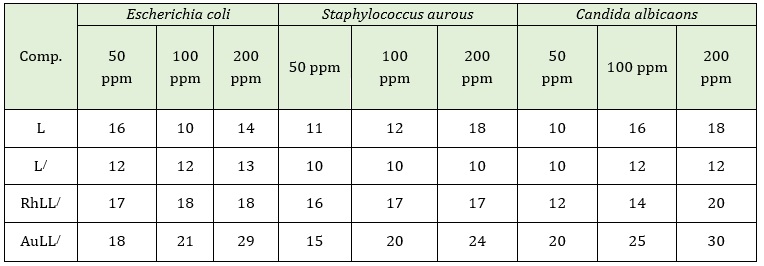

Figure 6: Antimicrobial activity of L, L/ and their inclusion metal complexes against selected microorganism
Conclusion
Two mixed ligands of sodium fusidate (L) and 2,2′-bipyridine (bipy)L/ were successfully synthesized by two chosen heavy metal complexes Rh(III) and Au(III) and diagnosed by chemical and physical tools to approaches for determining the mode of bonding and overall structure of these new complexes. The physicochemical and spectroscopic data propose that Rh(III), Au(III), and the ligands are brought together with a rigid configuration, and hence suggest that two ligands behaves as a bidentate one by oxygen atoms (L) and another by nitrogen atoms (L/), and also suggested the octahedral geometry for Rh(III) complex, while a square planner structure for Au(III) complex.
The biological activity of these free ligands and their inclusion metal (III) complexes were investigated in vitro against three selected microorganisms by using three concentrations and the outcome showed a good biological activity specifically the gold complex.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
Conflict of Interest
There are no conflicts of interest in this study.
ORCID
Mahasin F. Alias
https://www.orcid.org/0000-0002-3375-1797
HOW TO CITE THIS ARTICLE
Zahraa Q. Mahdi, Mahasin F. Alias. Synthesis, Characterization, and Antimicrobial Activity by Coordinated Metals Ions Rh+3 and Au+3 with Sodium Fusidate and 2,2/ Bipyridine as Ligands. Chem. Methodol., 2022, 6(12) 929-939
https://doi.org/10.22034/CHEMM.2022.355688.1591
URL: http://www.chemmethod.com/article_155569.html


