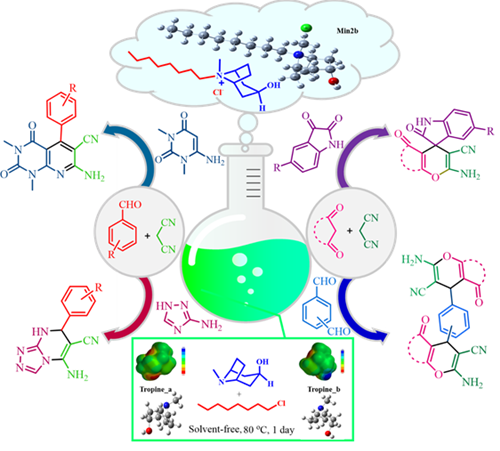
Introduction of a New Ionic Liquid Based on Tropine for the Acceleration of the Synthesis of Several Hetero-Aromatic Compounds and Investigation of Its Physicochemical Property and Antibacterial Activity
Pages 401-438
https://doi.org/10.48309/chemm.2024.452735.1789
Mahdi Ramezanpour-Touchahi, Masoumeh Mazloumi, Hossein Taherpour Nahzomi, Frahad Shirini, Hassan Tajik
Abstract In this work, we synthesized a new ionic liquid ([C8-Tropine].Cl) by combining tropine with 1-chlorooctane without the use of any solvent. Prepared ionic liquid was characterized utilizing Mass, TGA, CHN, FESEM, NMR, and FT-IR analysis techniques. Potential energy surfaces of different pathways and calculated energy barriers beside a few NMR chemical shifts were explored to determine the structure of the catalyst. According to the Curtin-Hammett principle, two different products can be obtained, named Min2a and Min2b, in a ratio of 0.26:1 which the structures of these products were determined by comparing the experimental and calculated NMR chemical shifts. The calculations were performed using the DFT (B3LYP) computational method with 6-31+G(d,p) and 6-311G(d,p) basis sets. The antibacterial activity of the catalyst was evaluated against Escherichia coli (Gram-negative) and Staphylococcus aureus (Gram-positive) using the agar well diffusion method. After the identification of the synthesized ionic liquid, it was utilized as an efficient catalyst for the synthesis of pyrido[2,3-d]pyrimidines, 1,2,4-triazolo[4,3-a]pyrimidines, bis-2-amino-4H-pyrane and spiro-2-amino-4H-pyran-oxyindole derivatives. These methods stand out for their convenient and uncomplicated catalyst preparation, minimal catalyst usage, straightforward work-up procedures, quick reaction times, and the ability to achieve good to high product yields under environmentally friendly conditions. Furthermore, the catalyst can be easily recovered and reused several times.