Syntheses and Structural Investigation of Two Novel Lanthanum Complexes with Salen Ligands Derived from 1,2-Diaminopropane and Ethylenediamine with 3-Methoxysalicylaldehyde
Pages 492-503
https://doi.org/10.48309/chemm.2024.458988.1798
Mohammad Hakimi, Mohammadreza Fotouhi Ardakani, Alireza Mazloumi Bajestani
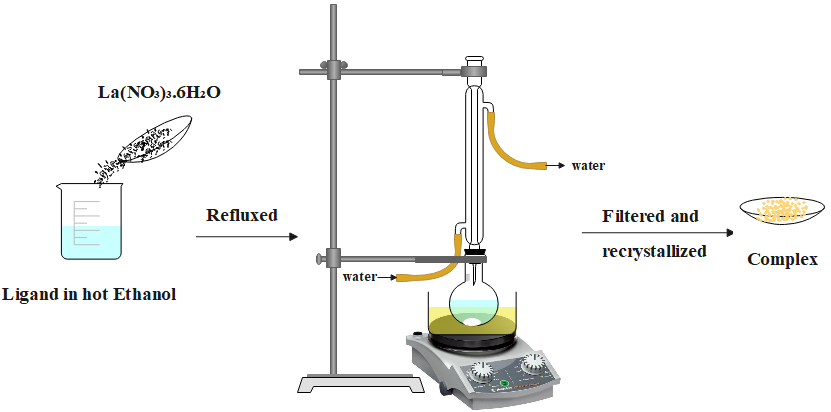
Abstract Recently, the chelation chemistry of Schiff base lanthanide (III) complexes has been widely considered. This work has concentrated on novel types of binuclear lanthanum complexes derived from two salen ligands including N,N′-bis(3-methoxysalicylaldehyde)-1,2-diaminopropane (H2La) and N,N′-bis(3-methoxysalicylaldehyde)-ethylenediamine (H2Lb). The salen ligands have been formed by the precipitation of ethylenediamine and 1,2-diaminopropane with 3-methoxysalicylaldehyde. Different analytical methods have been used for the characterization of prepared compounds in particular elemental analysis, UV-Vis, FT-IR, 1H-NMR, and 13C-NMR spectroscopy. The bidentate behavior of two salen ligands was suggested based on spectral studies. According to the spectral results, the proposed coordination of two salen ligands and their lanthanum complexes was approved.