Theoretical Investigation for the Optoelectronic Properties of Pyridyl-Fuoren-9 for Usage as Dye-Sensitized Solar Cells
Pages 776-788
https://doi.org/10.48309/chemm.2024.483222.1839
Sharif Abu Alrub, Suzan K. Alghamdi, Ahmed I. Ali, Rageh K. Hussein, Dina Ezzat, Sally A. Eladly
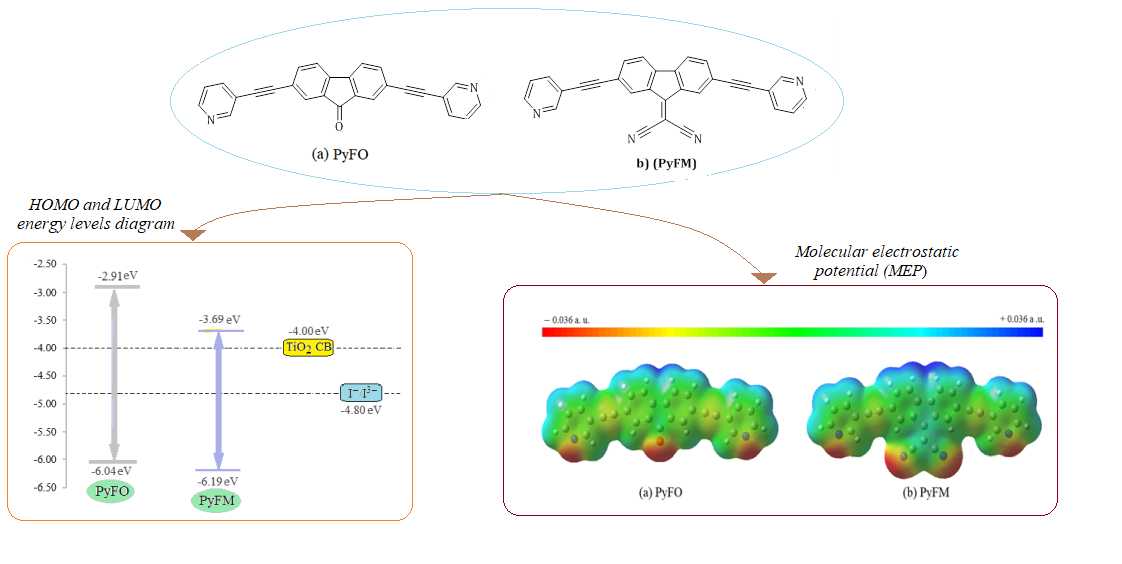
Abstract In this work, density functional theory and molecular dynamics calculations are used to explore the potential of pyridyl-fluoren-9 compounds for dye-sensitized solar cell applications. The study utilized two compounds: 2,7-bis(pyridin-3-ylethynyl) fluoren-9-ylidene) malononitrile (PyFM) and 2,7-bis(pyridin-3-ylethynyl) fluoren-9-one (PyFO). The substitution of a malononitrile group for the oxygen atom yielded PyFM from PyFO. PyFM exhibited smaller HOMO-LUMO energy gaps than PyFO due to the presence of the malononitrile group. The calculated quantum chemical descriptors of PyFM, such as hardness (η), softness (σ), and electronegativity (χ), revealed improved reactivity properties. The molecular orbital analysis identified a favorable intramolecular charge transfer between donor and acceptor components, classifying the studied molecules as D-π-A-π-D dye. The molecular electrostatic potential of PyFO and PyFM recognized their nucleophilic centers, which have significant potential for interaction with the electrophile atoms of the TiO2 surface. PyFM is predicted to exhibit high light-harvesting efficiency, as indicated by its broader, maximized, and redshifted absorbance peak at 597.02 nm in the calculated UV-vis absorption spectra. The estimated adsorption energies of PyFM dye pointed to a strong adsorption on the TiO2 surface. The study indicates that the performance of the pyridyl-fluoren-9 compound as solar cell dye can be enhanced by incorporating a malononitrile moiety instead of the oxygen atom in PyFO. This study encourages further exploration of pyridyl-functionalized fluorenes in dye-sensitized solar cell applications of pyridyl-functionalized fluoren-9 compounds in dye-sensitization solar cell applications.