An Electrochemical Sensor Based on SnSe Nanosheets and Ionic Liquids for The Selective Detection of N-acetylcysteine
Pages 158-171
https://doi.org/10.48309/chemm.2025.500065.1882
Peyman Mohammadzadeh Jahani, Reza Zaimbashi, Hadi Beitollahi, Somayeh Tajik
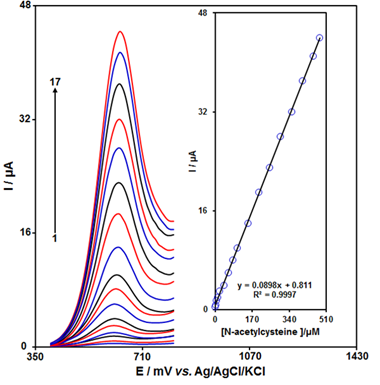
Abstract To detect N-acetylcysteine (N-AC), a voltammetric sensor was developed employing a carbon paste electrode (CPE) modified with SnSe nanosheets (SnSe NSs) and ionic liquids (ILs). Compared to the unmodified CPE, the electrochemical sensor (SnSe NSs/ILs/CPE) exhibited significantly higher current responses for N-AC oxidation. Using linear sweep voltammetry (LSV), cyclic voltammetry (CV), chronoamperometry, and differential pulse voltammetry (DPV), the electrochemical properties of the sensor and the oxidation behavior of N-AC were investigated. With a detection limit (LOD) of 0.02 µM (S/N = 3), DPV, a sensitive analytical method for N-AC quantification, showed a linear relationship between N-AC concentration and peak current spanning the range of 0.05 µM to 480.0 µM. In addition, the sensor demonstrated adequate stability and repeatability. With recovery rates ranging from 91.40% to 106.30%, it was effectively used to determine N-AC in actual samples.