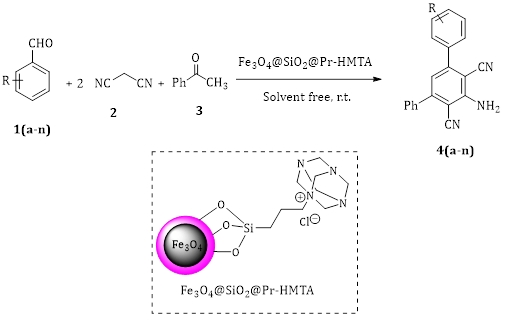
Application of Basic Magnetic Phase Transfer Nanocatalysts in the One-Pot and Multicomponent Synthesis of Poly-Substituted Aniline
Pages 403-416
https://doi.org/10.48309/chemm.2025.486122.1843
Hassan Hassani, Somayeh Heidary, Sayed Hossein Banitaba
Abstract Using a nano-phase-transfer catalyst (NPTC) to facilitate diverse organic reactions offers a promising approach to achieve mild, synergistic, and multi-component synthesis of valuable compounds under heterogeneous conditions. This study describes the synthesis of 3,5-diaryl-2,6-dicyanoanilines through a tandem cyclocondensation reaction using commercially available reagents and Fe3O4@SiO2@Pr-HMTA (hexamethylenetetramine) as the NPTC. The reaction takes place in water, a green solvent, and involves the condensation of acetophenone, malononitrile, and an aromatic aldehyde at room temperature. Notably, this methodology represents the first green, one-pot process for synthesizing poly substituted aniline derivatives using magnetic phase transfer catalyst in water.