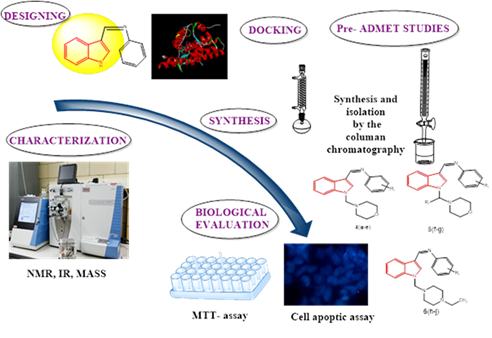
Design, Synthesis and Evaluation of Novel Phenyl-(1-morpholine-4-dimethyl/piperazin-1-ylmethyl)-1H-indol-3-ylmethylene Amine Derivatives Against Breast Cancer Cells
Pages 851-866
https://doi.org/10.48309/chemm.2025.517119.1938
Ranjit Vinayak Gadhave, Rutuja Sadashiv Dhongade, Shweta Dilip Raut, Mayuresh Abhay Shastri, Preeti Prashant Mehta, Yogita Sachin Ozarde
Abstract The indole derivatives, phenyl-(1-morpholin-4-yl methyl/piperazine-1-yl methyl)-1H-indol-3-ylmethylene amines were designed for interaction with progesterone receptor. The molecular docking was performed on progesterone receptor using crystal structure with PDB ID: 4OAR. Out of 139 designed molecules 10 molecules showed good binding interactions withGlu-695, Asp-697, and His-770amino acid residues, which are vital for required binding orientation of target protein binding cavity and are similar to interactions of reference standard Sunitinib. The combination of hydrogen bonding, hydrophobic, and ionic interaction confirmed the robust nature of the ligand-protein binding and docking score ranged from -7.5 to -9.9 kcal/mol. IBMA-2 demonstrated the most potent binding affinity of -9.9 kcal/mol. The synthesized compounds were evaluated on MCF-7 cell line for anticancer activity. In the MTT and cell apoptosis assays IFMA-4 and IFMA-5 showed significant activity with IC50 values of 37.13±0.64 μg/mL and 40.94±0.86 μg/mL, respectively, compared to reference standard Sunitinib having IC50 value 30.05±0.96 μg/mL. Molecular docking study aligned with the biological activity results. The study highlighted the role of progesterone receptor in treatment of hormone receptor positive breast cancer. The synthesized compounds displayed optimum biological activity. Therefore, these can be treated as lead nucleus for further structural modifications.