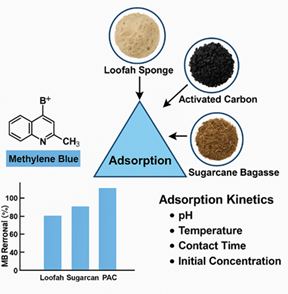
Chemical Characterization and Adsorption Kinetics of Methylene Blue: A Comparative Study of Activated Carbon and Plant-Based Bioadsorbents
Pages 994-1015
https://doi.org/10.48309/chemm.2025.529712.1972
Jana Rammal⸸, Mohamad T. Raad⸸, Hassan El Said⸸, Salma Mhanna⸸, Zaher Abdel Baki, Dalia El badan, Mohamad Alameh, Akram Hijazi, Ismail Hijazi
Abstract The search for efficient, low-cost, and sustainable adsorbents is vital to tackling global water pollution. This study compares powdered activated carbon, loofah sponge powder, and sugarcane bagasse powder for removing methylene blue (MB) from water. Physicochemical characterization included pH, moisture and ash content, FTIR spectroscopy, iodine number, particle size, and phenol adsorption to understand the adsorption mechanisms. Batch experiments examined key factors: pH (2.5–11.1), temperature (5–55 °C), contact time (10–150 min), dye concentration (25–400 mg/L), and adsorbent dosage (25–300 mg). Under optimal conditions (20 mg dosage, pH 8, 40 min), PAC showed the highest MB removal efficiency (99.85%), attributed to its high microporosity and oxygenated surface groups. Natural bioadsorbents—loofah and sugarcane powders—also demonstrated strong removal efficiencies (96.64% and 97.53%), highlighting their potential as biodegradable, low-cost alternatives. Compared to literature reports, these bioadsorbents outperform coir pith (76.92 mg/g), pine cone (84.32 mg/g), and zeolite (71.28 mg/g), and greatly exceed activated alumina (55.30 mg/g) in MB adsorption capacity. This demonstrates the superior performance of loofah and sugarcane powders studied here. A strong linear correlation (R² ≥ 0.998) between phenol concentration and UV–Vis absorbance confirmed phenol adsorption as a reliable proxy for adsorption behavior. Adsorption was closely linked to surface functional groups, pore structure, and particle size, supported by FTIR and iodine number data. These results highlight bioadsorbents’ suitability for decentralized or resource-limited water treatment, offering sustainable, affordable solutions. Overall, this study supports integrating eco-friendly, bio-based adsorbents into scalable water purification technologies, advancing sustainable environmental remediation and clean water access.

![Novel Imidazo[1,2-<em>α</em>]Pyridine Hybrids: Synthesis, Antioxidant Activity, Cytotoxicity against Cancer Cell Lines, and <em>In Silico</em> Docking Analysis](data/chemm/coversheet/121752391805.png)