A Short Review on Supercritical Fluid Extraction: A Key to Good Performance
Pages 1-14
https://doi.org/10.48309/chemm.2026.532286.1977
Susan Khosroyar, Nadia Sahebjamee, Farzad Kaj
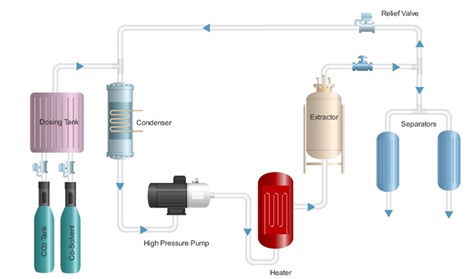
Abstract The isolation of components of interest is important in many industries because it impacts the quality of the product, the economics of the process, and the environment. Supercritical fluid extraction (SFE) is a modern method of extraction that is more versatile and eco-friendlier than traditional methods, as it is highly selective and offers fast mass transfer along with high solvent recovery. It is used in the pharmaceutical, nutraceutical, food processing, cosmetics, and natural products industries. In this review, the basic principles of SFE and the important operating parameters—temperature, pressure, solvent flow rate, supercritical fluid type, particle size, extraction time, and feed moisture content—are discussed concerning extraction yield and efficiency. Both thermodynamics (phase behavior, solubility, and density effects) and kinetic factors (mass transfer rates and diffusion limitations) are included to discuss how these factors affect the driving force of the process. It is shown that with proper selection of parameters, yield, selectivity, and even sustainability can be improved, thereby making SFE more competitive for the recovery of valued compounds. This information provide a useful guide to help researchers and industry experts aiming to design, scale up, and apply effective SFE systems across different uses.