Efficient Solvent-Free Synthesis of Imidazoles Using 2-Oxopyrrolidinium Bisulfate as a Novel Acidic Ionic Liquid Catalyst
Pages 223-233
https://doi.org/10.48309/chemm.2026.547253.2013
Dariush Felekari
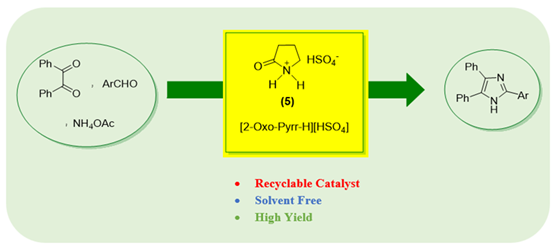
Abstract The development of novel ionic liquids (ILs) with dual functions as catalysts and green reaction media continues to attract considerable attention in sustainable organic synthesis. In this study, the synthesis of 2-oxopyrrolidinium bisulfate, a new, efficient, and environmentally benign acidic IL was reported. Its catalytic performance was evaluated in the one-pot, three-component synthesis of imidazole derivatives through the condensation of benzil, aldehydes, and ammonium acetate under solvent-free conditions. The protocol afforded the desired imidazoles in high to excellent yields within short reaction times, highlighting the efficiency of the system. Beyond its catalytic activity, the IL exhibited excellent recyclability and could be reused for at least four consecutive cycles with minimal loss of performance, confirming both its stability and practical applicability. The superior performance of 2-oxopyrrolidinium bisulfate compared to conventional acid catalysts can be attributed to its strong Brønsted acidity, hydrogen-bonding ability, and homogeneous nature, which collectively facilitate efficient activation of substrates under mild, solvent-free conditions. Due to its ease of preparation, operational simplicity, non-chromatographic product purification, and low environmental footprint, this IL represents a promising addition to the library of task-specific catalysts. Moreover, the methodology aligns with the principles of green chemistry by eliminating volatile organic solvents, minimizing waste, and enabling catalyst recovery. These findings not only establish 2-oxopyrrolidinium bisulfate as a powerful medium for imidazole synthesis, but also suggest its potential applicability in a broader range of heterocyclic and multicomponent transformations.