Molecular Dynamics Insights into GO–SiNP Nanocarrier Interactions with Stroke Biomarkers: Toward Rational Biosensor Engineering
Pages 355-369
https://doi.org/10.48309/chemm.2026.564269.2063
Mohamad T. Raad†, Hassan El Said†, Josiane Alhage†, Abdallah Al Jammal, Hussein Hassan, Mohammad Y. Chreif, Mostafa Al Zein†
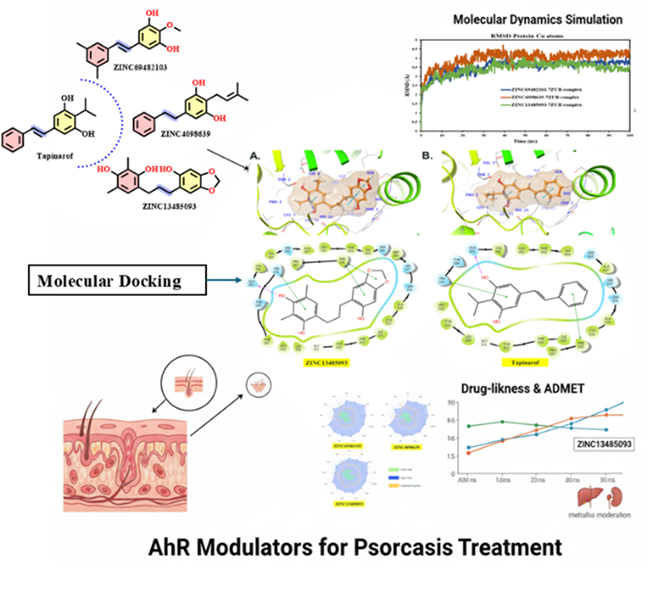
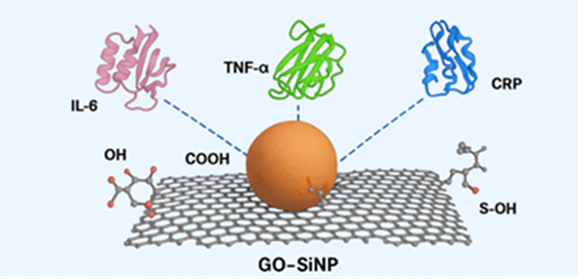
Abstract Early stroke detection is limited by a lack of sensors that can capture early inflammatory biomarkers. interleukin-6 (IL-6), tumor necrosis factor-α (TNF-α), and C-reactive protein (CRP) rise rapidly during neuroinflammation, serving as key early indicators. This study used all-atom molecular dynamics (MD) simulations (100 ns, triple replicates) to analyze interactions between these cytokines and silica-functionalized graphene oxide (GO–SiNP), directly comparing it to pristine graphene oxide (GO) and contextualizing findings against literature reports for SiNP, AuNP, and PEGylated carriers. Within our simulations, GO–SiNP provided superior stabilization versus GO alone, showing lower Root-mean-square deviation (RMSD) values (0.18–0.26 nm) and 20–35% reduced residue flexibility. The hybrid surface formed more persistent hydrogen bonds (6.5–11.0 on average) with the cytokines, supported by sharp radial distribution function (RDFs) peaks at 0.25–0.32 nm, indicating strong, short-range polar coordination with silica oxygen atoms. MM-PBSA calculations confirmed significantly stronger binding to GO–SiNP (−158 to −172 kJ.mol⁻¹) than to GO (−128 to −140 kJ.mol⁻¹). Crucially, silica functionalization did not hinder graphene's aromatic activity, maintaining 9–11 π–π contacts per cytokine versus 5–7 on GO. The binding affinity order was consistently TNF-α >IL-6 >CRP. These results demonstrate that GO–SiNP enables a dual-mode stabilization mechanism—combining polar anchoring with π-π interactions—unachievable by its individual components. Compared to conventional nanocarriers optimized for a single interaction type, this hybrid design represents a distinct, integrative strategy for robust biomarker capture. This work provides a molecular framework for designing advanced biosensors for early stroke detection.

![Preparation of [1,2,4]Triazolo[1,5-<em>a</em>]Pyrimidines Accelerated by Magnesium Schiff Base Complex Immobilized on Magnetite Nanoparticles](data/chemm/coversheet/91767772449.png)