Document Type : Original Article
Authors
- Bahman Fazeli-Nasab 1, 2
- Laleh Shahraki-Mojahed 3
- Zahra Beigomi 4
- Maryam Beigomi 5
- Anahita Pahlavan 6
1 Research Department of Agronomy and Plant Breeding, Agricultural Research Institute, University of Zabol, Zabol, Iran
2 Department of Biotechnology and Plant Breeding, Faculty of Agriculture, Ferdowsi University of Mashhad, Mashhad, Iran
3 Department of Biochemistry, School of medicine, Zabol University of Medical Sciences, Zabol, Iran
4 Department of Physical Education, Zahedan University of Medical Sciences, Zahedan, Iran
5 Department of Food Science and Technology, Zahedan University of Medical Sciences, Zahedan, Iran
6 Department of Horticultural, Faculty of Agriculture, University of Zabol, Zabol, Iran
Abstract
In recent years, environmental concerns and food safety in developing countries are the most important issues. Pesticides are vastly utilized in farming to improve crop production and quality and to reduce energy costs. These pesticides are biodegradable chemical compounds that are generally applied in farming to control pests and promote agricultural production. However, their excessive use, even in low concentrations, may cause serious health problems and environmental pollution. Therefore, a great deal of the research has focused on methods used for determining the presence of pesticides in various food matrices, according to this proposing sensitive diagnostic methods is essential for reliable quantification of pesticides availability. Meanwhile, rapid detection technologies which are among the most important tools in the analysis of food products are required to be assessed for analysis of residual persistence of pesticides in food and agricultural products to effectively control their quality and safety. In this article, it is attempted to provide a complete review of some detection methods including HPLC, HPTLC, GC/ MS/ MS, CE-DAD, LPME, SPME, LLE, DLLME, AChE, SERS, LC/ Tandem/ MS/ MS, MSPD, luminescence chemistry, safety assessment, and Meisel chromatography electrosynthesis used to identify pesticide residues in different food samples.
Graphical Abstract
Keywords
Main Subjects
Introduction
The Sudden burst of the world's population has made food demand the most significant concern in both developed and developing nations. Since most of these people are vegetarian, their diet depends greatly on agricultural products to meet their daily nutrient needs, so the developed countries are more comfortable in producing such crops [1].
Due to the presence of insects and mites (around 9,000 species) worldwide, it is possible to control optimal production of agricultural products for human consumption, so in improving crop production of agricultural products, monitoring harmful insects is of great significance [2]. Besides, agriculture consists of the major livelihood of the world's population (over 60%). The rapid growth of the world's population is the main reason for food insecurity at this evergrowing rate. Meeting the needs of the global population requires using more scientific approaches to increase the yield of agricultural products with higher sustainability. Also, producing desirable food at the global level along with controlling pests is vital since more than 50% of food production is lost annually by pests. In attempts done to increase and promote agricultural productivity, pesticides have become an important tool used as crop protectors. Chemical pesticides which can control different pests and plant diseases play a significant and potential role in increasing agricultural yield [3-8].
The term pesticide covers a wide range of compounds such as pesticides, fungicides, herbicides, rodenticides, mollusks, and other lethal matters. Pesticides and herbicides are the second and the third most popularly used pesticides in the world, respectively. Organochlorine pesticides are applied successfully to control contagious agents, yet after the use of organochlorine insecticides was banned in the 1960s, other synthetic pesticides like OP, CM, and pyrethroids were introduced [3].
Pesticides are one of the types of neurological compounds that are broadly utilized to ensure the high yield of the product due to the reduction of pests and eradication of diseases, but they are additionally highly poisonous to living organisms. Particularly in developing countries, many serious food safety problems have been raised, as pesticides are widely used and not adequately monitored [9].
Pesticides have numerous usages for pest monitoring in farming, manufacturing, and human habitats. Although crop production has increased significantly by using such pesticides, their excessive use causes pollution in the environment, food, water, and farming outcomes. The appearance of pesticide residuals in human food like milk is a warning symbol requiring immediate implementation of a close monitoring system with eradication treatments [3]. Food safety problems are high due to the residual pesticides in fruits and vegetables [10].
Annually, about 6.5 billion pounds of pesticides are consumed across the world. Here, the most concerning ecological point is the fact that pesticides are reciprocally and continuously transferring from abiotic to biotic medium, and in turn, leave their negative effects on both environments. In addition, studies have shown that more than 98% of insecticides and 95% of sprayed herbicides are used for other purposes as well which include non-target species, air, water, soil, and food [11].
Plant protection products have been used on a large scale since the 1940s. Active substances included in the commercial preparations allow increasing the yield from arable lands. Over the years, new pesticides have been introduced to replace those proven to be toxic to animals and humans. Currently, pesticides are subject to stringent legal regulations defining the maximum residue concentration in food; consequently, it is essential to control their content in food [12].
In general, pesticides are designed to leave lethal effects on the targeted pests rather than the non-target population. Yet, because of their sustainable nature, pesticide residuals remain longer in the environment, and eventually, enter the food chain. Residues of pesticides may penetrate the food chain by the atmosphere, earth, and water. They influence the environment and make some health difficulties for living organisms. Pesticides can cause cancer, neurological disorders, infertility, immune and respiratory diseases [3]; hence, continuous monitoring of pesticides left in food and the environment is essential to a certain compliance with legal restrictions in a comprehensive way.
History of food exposure to pesticides
Residues of chemical compounds in nutritions cover a vast range from natural toxins, chemical compounds AAin agriculture, bio-industrial pollutants to chemical materials caused by food processing and packaging. The case of not using pesticides, crop loss in pre-and post-harvest stages is estimated to be about 46 percent in some countries[13, 14]. In Iran, out of 800 kinds of pesticides used in the world, 211 kinds of chemical compounds with various formulations and applications are recorded [15].
More than 80 percent of pesticide residues cause serious harm to humans, especially in children [16]. Moreover, about 20 percent of poisonous plant pest control may cause cancer so, due to health and economic consequences, proper monitoring of residual toxins available in foods is necessary. Thus, monitoring programs of pesticide residues in food are continuously performed in many countries to ensure maximum allowable pesticide residues and intake in the human diet [16]. The World Health Organization (WHO) has also classified all poison plant pest control in terms of hazardous active toxic compounds and LD50 into 4 sub-categories: Severe, high, medium, and partial [17].
Since ancient times, pesticides, which have been regarded as a means for controlling insects, were used as the main ingredient. The widespread use of plant protection compounds and chemicals began in the mid 19th century with the invention of DDT (Figure 1), which has the highest contact toxin for various insects [18]. The second half of the 20th century was the ending point for DDT, since it not only had any effects on pests but also it left serious side effects on various organisms. Later, DDT was finally replaced by organophosphate composites [19], which include a wide range of compounds with significant advantages in farming, public health, and the inner field [20].

Figure 1: 2D and 3D chemical structure of DDT (source: wikipedia.org)
Currently, researchers use different ways to determine the pesticide residual content in agricultural products by recognizing and measuring numerous pesticides by applying gas chromatography (GC) methods with the NPD, ECD, FPD detectors [21-23].
Methods of identifying pesticides in environmental and agricultural samples
Although pesticides are widely used in agriculture, they have greatly contaminated different parts of the environment and their residues can be found in environmental and agricultural samples. There are numerous reports indicating the fact that the increasing rate of using pesticides for agricultural purposes has caused a clear effluent in the environment which can be a major threatening risk to the health of all living organisms [24].
Contamination of a number of vegetables and fruits with a number of fungicides has been investigated using the IFS and risk factor (R) to evaluate the health hazards of pesticides to users. Although difenoconazole and primidone in vegetables were more than the standard levels, carbendazim, difenoconazole, and tebuconazole were the most popular pesticides found in farming products. The fact that IFS of all pesticides in all farming products is < 1 also represents the immunity risk of pesticide residuals. The levels of R-difenoconazole and primidone in farming products have been moderate, so it is suggested to consider them within the main control factors to be monitored continuously to ensure individual health [25].
Therefore, it seems urgent to constantly monitor pesticide residuals in food and environmental samples to get ensured of their accurate and comprehensive compliance with legal restrictions [26]. In this regard, numerous analytical and quantitative methods such as MS, GC, and HPLC, capillary region / MS electrophoresis, chemiluminescence and immunoassay are developed to detect organophosphorus (OP) compounds both in environmental, and agricultural samples [19]. Recently, a great number of scientific articles have been published on chromatographic techniques, which are specified to represent the ability of chromatographic methods in identifying pesticide residuals in food as well as in environmental samples.
More recently, numerous advances have been made in the identification and quantification of pesticides in various matrices. Although banned for their health harms, OP pesticides are still extensively employed in farming. Visual detection or colorimetric pesticides using various nanostructures are significantly exploited to monitor the accumulation sites of OP pesticides [3]. Nanotechnology is also extensively explored for the development of analytical tools. Color or visual tracking of pesticides using metal nanoparticles is one of the simplest and most cost-effective ways to detect several types of pesticides residue. Moreover, high-tech visual detection techniques use metal nanoparticles, which mainly act based on the phenomenon of localized surface plasmon resonance to identify several residual toxins in a large population of samples [3, 7, 27].
Generally, pesticide detection methods include GC, GC-MS, HPLC, and HPLC-MS. Moreover, some other analytical methods are described in the research, which are based on biosensors, spectrophotometry, and CE-DAD [25].
Identification of pesticides using chromatographic methods
The most crucial step in the characterization of nutrient and bioactive molecules is to select the appropriate analytical strategies. Although spectral protocols usually allow for the identification and quantification of food ingredients; the particular chemical composition may not be fully recognized in most samples. To solve this problem, spectrophotometry is commonly used to assess antioxidant capacity [28]. Yet, more specific knowledge on chemical composites and further analysis methods, such as chromatography and MS, are required to offer a better picture of the structures of mixtures in food forms. In this regard, recent improvements in technology and innovations in analytical tools have offered more complex (quantitative and qualitative) assessments of the chemical composition of nutrition and health in food [29].
Chromatography is commonly used to analyze natural foods and related derivatives. Such chromatographic methods can give a very powerful separation capability since chemical materials found in multiple extracts can be separated into somewhat easy parts (subgroups). Moreover, the latest analytical approaches acting as hyphenate spectroscopy and chromatography (for example, CE-DAD, HPLC-DAD, high and gas chromatography, GC- Mass, HPLC-MS, and HPLC-NMR) may give supplementary spectral knowledge. These techniques are really useful in online structural clarification and qualitative analysis [30, 31].
Various methods like MS simple sequential detector of ionization electron-chemical, selectively following sequence (GC / MS / MS), HPLC, and HPLC detector MS with a dual focus (LC / Tandem / MS / MS [32-34], MS, and GC are also well specified to detect and quantify these OP compounds in environmental and agricultural samples. Although these methods are sensitive, specific, and reproducible, they are time-consuming and extravagant and require hiring advanced tools and qualified experts [24, 35]. Therefore, simple, fast, and cost-effective methods are always considered in identifying pesticide residues.
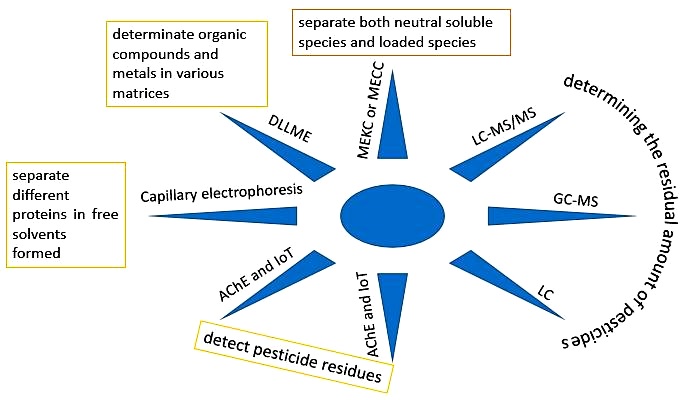
CE-DAD
CE was initially used to represent the separation of different proteins in free solvents formed as continuous bands of a tube. Mineral ions, proteins, nucleic acids, and microorganisms can be disintegrated by using CZE and CIEF. Later, the accessibility to capillary pipes of melt silica covered with polyamide with the inner diameter of 75-100 μm along with a sensitive absorption detector (with the inner diameter within µ level) made for HPLC acted as a useful means to pave the way for the development of commercial applications of CE. The less inner diameter can also remove band broadening which is caused by transport, whereas due to its features such as plug flow and electro-osmotic flow (EOF), it can also make the possibility to reach 100,000 efficiency of the theoretical sheets [32, 36].
CE is a special case of applying an electric field to the separation of components in a compound. The difference between a CE with other forms of electrophoresis is that it is applied within the borders of a thin pipe. To understand the behavior of the molecules affected by the electric field in the capillary, it is necessary to get familiar with phenomena caused by capillary geometry. CE is used in identifying DNA sequence, extraction of proteins, peptides, chiral compounds, medicines, mineral ions, self-organization, identification, and separation of nanomaterials, and criminal analysis. The most important kinds of CE include the following instances [32, 36]:
Regional CE
Regional CE is the simplest form of CE in which it is required to fill the capillary with a running buffer. In this diagram of the CE, the ionic solved species transfer with different speeds (clarified by the ratio of load/ mass) to create the distinct regions in the running buffer. Using Electroosmotic flow (EOF), it is possible to separate a great number of cation/ anion dissolved species, yet the neutral species which move by the osmosis electron flow remain unseparated [37].
Biopharmaceuticals are prone to physical and chemical instability due to their inherent physicochemical complexities, so one of the important dimensions in the quality control of these drugs is their stability studies. Undoubtedly, stability studies will not be possible without reliable analysis methods indicating stability. Capillary electrophoresis is one of the most powerful analytical methods and in parallel with extensive advances in the production of biopharmaceuticals in the last two decades, extensive efforts have been made to provide new analytical methods based on capillary electrophoresis to control the quality of these drugs [38-41].
MEKC or MECC
MEKC or MECC is a combination of electrophoresis and chromatography which can separate both neutral soluble species and loaded species. A great advantage of this CE is that as the concentration of some surfactannts reaches critical micelle concentration (CMC), the micelles are created which helps the separation of the neutral species. These spherical micelles contain hydrophilic tails of surfactant molecules that tend toward the center and loaded heads, inclining toward the exterior of the buffer. After applying a potential field across the capillary, these closed micelles are loaded toward their loads and immigrate so that the dissolved species between the micelles and liquid phase are divided which in turn, conserves some species (differential retention) and isolate the dissolved species. Using different types of surfactants can change micelle physical nature and micelle sensitivity as well. Some applications of micelle’s electro synthetic Chromatography are aminoacids' separation, heavy metals, nucleotides, vitamins, and drugs [25,32,36].
Today, using CE- based methods for identifying and determining various analyses such as medicine with treating purposes and illegal drugs in biologic samples and underground products is quite popular. There are also other widespread ways like GC and HPLC which are useful in clarifying analytes. The priority of CE over the two other earlier-mentioned methods is that it needs the least number of samples for conducting a test. It is possible to state that the best test results are obtained when a combination of CE and either GC or HPLC is used. Practical usage of CE, regarding the scarcity of the samples it requires, is Gunshot Residue, which is collected on human skin [42].
GC or HPLC
Accurate determination of pesticide residues in real samples can be done mainly based on standard reference methods such as GC or HPLC [10]. These two methods have the same disadvantages, such as time-consuming steps, complex operations and high costs. Therefore, it is necessary to provide simple, fast and low-cost methods for determining the residues of pesticides. Accordingly, there are reports of studies on specific tracing of pesticides and CM based on inhibition of ACh activity [43]. The main drawback of using a spectrophotometer [10] or biosensors [44] is that they focus on enzymatic analysis. However, these methods have simplified the testing process and reduced the determination time. But they still need electricity and equipment, which makes it difficult to measure quickly on site. In addition, there are still some problems in biosensors that need to be further investigated and improved, such as poor reproduction and poor stability to detect pesticide residues in real samples [9].
Today HPLC is the most widely used analytical method [45]. It is known as a standard strategy in food characterization. it has some shortcomings such as high consumption of natural solutions, long analytical time, and the high price of tools [46]. A marker is a chemical form of raw substances, preparations, beverages, or foodstuffs which are used for quality control properties or purposes, especially in cases where nutrients or active ingredients have not been previously identified [47].
The residual amount of different kinds of pesticides was evaluated by MC-MS in some vegetables supplied in the central fruit and vegetable business of Tehran in 2005. It was concluded that of the 30 selected samples, 53.33% had pesticide residuals. Toxins including Endosulfan (I, II, sulfate), Chlorpyrifos, and Phosalone were significantly recognized in cucumber and tomato samples. The average recovery of toxins with this method was good (<60%). The residual levels of toxins detected in the contaminated examples were lower than the maximum approved by FAO / Codex and the European Union. None of the unauthorized toxins such as DDT and HCH was present in the samples [15].
HPTLC Method
TLC and HPTLC are the preferred methods for identifying carbosulfan (an insecticide and member of the carbamate family), the leading cause of most human poisoning. Carbosulfan produces more intense color compounds than other organochlorines, organophosphorus, and pyrethroid insecticides but does not interfere with biological substances (amino acids, peptides, proteins, fats, etc.) [48].
SPME and LPME
SPME and LPME play a significant function in pesticide analysis in various matrices. Hollow liquid phase microextraction is a simple, inexpensive, and fast method of extraction which involves sampling, extraction, and pre-concentration simultaneously, and offers excellent cleaning for complex matrices. Among different modes of LPME, micro-extraction of HF-LPME gives better sensitivity and accuracy, since the organic phase which is protected by a layer can prevent the dissolution of the organic phase compared to SDME [49].
LLE
LLE is one of the fastest ways used for breaking down residual food pesticides. LLE, a kind of extraction done by direct partitioning with an inflexible solvent, also uses furanolites in solutions or liquid samples [50].
Today, due to the high cost and harmfulness of organic solvents to the environment, many attempts have been made to replace new systems in industrial applications. Recently, two-phase aqueous systems have been put into operation for this purpose. These systems do not require the use of organic diluents that are typically toxic, volatile, and flammable. The cost used in aqueous biphasic systems is lower. Separation between phases occurs faster in these systems and the interface between the two phases seems clearer [51-53].
DLLME
Environmentally, due to its simplicity, speed of work, determination of organic compounds and metals in various and low consumption of solvents and reagents, DLLME has become a popular sample preparation method. This has attracted various scientists working in science, so it has progressed considerably since its introduction in 2006 [54,55]. This method includes hybrid use of different types of IL-DLLME such as META IL-DLLME, in-situ MR-IL- DLLME, and MIL-DLLME (Figure 2) [54].

Figure 2: Rapid detection methods of pesticides residues in vegetable foods
DLLME, compatible with current trends in modern decomposition chemistry, is simple, inexpensive, and environmentally friendly, and can provide a great deal of enrichment in a wide range of acceptor/ donor stages. The most significant DLLME parameters are the selection of extraction conditions and the selection of dispersed solvents for analysis extraction. A proper dispersion solvent should be mixed with both the extraction step and water to produce a cloudy solution which in turn, increases the interaction between the two steps, and provides high extraction efficiency [56].
However, since there are some potential faults in DLLME that limit its use in certain contexts, various attempts and recommendations have been made to improve this method. Some of these frequently-mentioned problems are limitations of extraction solvent (having a higher density than water for simple phase separation) and formation of a cloud solution in presence of a dispersed solvent. Another problem is related to its dependence on dispersed solvents [54,57].
Acetylcholinesterase (AChE) biosensor
GC, LC, or similar compounds such as GC-MS or LC-MS / MS are traditional analytical methods used for identifying and determining the residual amount of pesticides [58, 59]. Their popularity is due to the fact that they are inexpensive, not laborious, and convenient. Besides, they are unable to share information and control remotely, so they are not suitable for rapid detection and tracking of agricultural products. Biosensors can act as a cost-effective screening device [60, 61].
Along with the sudden burst of smartphones and wireless technologies and sensor technologies which have globally become an essential tool for everyday life, the future wave of connected devices, appliances, sensors, meters, and numerous other "things" represents the next generation of an overly connected world: The Internet of Things (IoT) [62].
AChE and IoT biosensors are today used to detect pesticide residues and track agricultural products (Figure 3). The system is designed to extend the benefits of pesticide residue detection history mainly due to its ability to remotely control, process, and share data and to track agricultural products using tracking devices (hypogynous computers) at detection sites. This diagnostic data which is obtained from tracking devices is further collected, processed, and analyzed by Epigynous computer to extract useful information such as detection time, production sites, detection samples, residual pesticide residues, and diagnostic inspector, which are effective factors playing roles in protecting quality and safety [60].

Figure 3: Architecture and of prototype the Detection System [60]
Using a screening chip for contaminated materials
Before identifying individual pesticides, some rigorous assays are built using antibody-coated membranes to act according to the high specific immune response. However, in practice, there are farmers in some cities who use different types of pesticides, so it is impossible to predict the exact types of pesticides. It makes selecting a membrane coated with the right antibody difficult to detect the unknown type of pesticide. In terms of food safety monitoring, development of a wide range of sensitive screening tools will have plenty of significant practical applications for identifying the level of damage between different pesticides in food [63, 64].
In a study, a rapid visual screening card was developed to identify remnants of pesticides containing membranes coated with enzyme and litter solution. The mechanism of the image discoloration in the test card is based on inhibition of Ache activity by pesticides and enrichment of the chemical on the membrane surface which is caused by its rapid evaporation [9]. Similr membrane chips are created based on enzyme reaction thiocholine and dithiobis-2-nitrobenzoic acid, which can change color from white to yellow on chip surface [65].
A sensitive visual screening card (chip), designed to identify pesticide residues, consists of an enzyme immobilized membrane with a suitable concentration of litter solution. Comparison of PVDF and Nymondrane Hybond in AChE uptake showed that the latter acts more suitablly as an enzyme carrier. The parameters of immobilization time, temperature, concentration, and volume of the enzyme were optimized for immobilization of AChE onto on the membrane chip. Moreover, the concentration of the substrate solution, the setting time, and the color development time were optimized to confirm the optimal conditions of the method. On the other hand, the results of pesticide residue detection in real food samples of fruits and vegetables showed that this visual screening card has high sensitivity, good reproduction capability, and stable storage properties. These features clarify its great potential for practical application in rapid determination of pesticide residues. The measurement results were therefore in line with the formal GCeMS method that showed the ability of the prepared assay system to screen OP and CM pesticides residues in food before formal quantitative analysis, especially in case of rapid sensitivity determination [9].
Surface Enhance Raman Scattering (SERS)
Efficient extraction of targets from complex surfaces is vital to technological applications ranging from monitoring environmental pollutants to analyzing the effects of explosives and pesticide residues. The SERS is used directly for rapid extraction and identification of pesticide molecules. The new substrate is made by decorating a commercial strip with colloidal gold nanoparticles (Au NPs) that provide simultenous SERS and "sticky" activities of the adhesive. The application of the SERS tape demonstrates a simple and lively "pulping and peeling" approach by directly extracting pesticide residues from fruits and vegetables. Strong and recognizable SERS signals ensure detection of various pesticide residues such as parathion methyl, tyram, and chlorpyrifos in real samples with complex surfaces like green vegetables, cucumbers, oranges, and apples [66].
SERS, which acts as an effective and sensitive tool, produces extreme Raman signals at the designed levels of absorbing molecules to offer various fields such as biology, biotechnology, internal security, and food security with different applications [66].
MSPD
MSPD which has a wide range of possibilities for analysis of solid, semi-solid, and highly viscous matrices, is a method used for mixig food natrix with absorbants through mortar and pot, which are transferred to a tiny column. The analytes washed with a small amount of a suitable solvent are more effective in eliminating interferences in food matrix. MSPD methods can increase extraction performance, reduce solvent consumption, and reduce extraction to purify samples in solid, semi-solid, and liquid conditions. However, the main limitation of this method is manual application of all its steps [50].
Machines designed to separate contaminated grains
In commercial applications, most seeds suspected to contamination with aflatoxins are isolated by removing stained or damaged seeds by insects using machines operating on a variety of factors. It is estimated that 66% of contaminated peanut kernels were removed by standard processing and isolation programs. Aspergillus flavus also secretes a compound called kojic acid which after reacting with brain tissue radiates BGY itself under the influence of ultraviolet light. This property is used to apply a machine for other products [67,68].
A high-speed separator, operating at a speed of 360 beats/ min., is designed to examine each peanut individually. Such devices are also designed to isolate possibly infected pistachios [69].
Organochlorine toxins
Organochlorine toxins which are among the most important kinds of organic toxins cover a wide range of pesticides. Since ancient times, these toxins have been widely used against malaria as well as various pests and insects, and due to their high impact, are still frequently used in various cases. This group of toxins can easily spread in nature and enter human life cycle in various ways. Due to the cumulative properties of these toxins in tissues and biological fluids which are rich in fats, some of their residues can be traced after years. Residual symptoms of these toxins in breast milk can be a clear qualitative and quantitative indicator for presence of such insecticides in the human body [70].
Humans have long used a variety of chemicals to repel pests, yet in turn, these substances have caused severe and irreparable damage to both environment and balance between its ecosystems and the health of biospecies. Among chemical compounds, organochlorine pesticides have caused more concern for their carcinogenic effects and the potential for causing delayed neurotoxicity. Dichlorodiphenyl trichloroethane was made by a German chemist in 1847. Later, an Austrian chemist, Pavel Müller, was awarded the 1948 Nobel Prize in Medicine for his discovery of the properties of this compound eventually known as DDT. As the first synthetic insecticide acting against plant pests and disease carriers, DDT has many positive and negative effects on human health. The effect of this toxin is so profound that lots of modern concepts in pest control and vectors like the theory of integrated pest management have been proposed to reduce its destructive effects, and even eliminate its application. Introducing this substance was among the most interesting and challenging discoveries of human knowledge. In the northern regions of Iran, women and children are more at risk of getting poisoned by chemical toxins, including organochlorine compounds, mostly because of greater involvement in agricultural activities [71].
In 1994, residues of organic chlorine toxins in 40 samples of breast milk in Tehran were evaluated using GC method, and it was concluded that the average residues of 5 selected toxins, linden, arochlor, pp DDT, op DDT, and pp DDE in breast milk were 02.23, 66.110, 4.2, 3.4, and 96.14 ppb, whereas the average standard amount of these toxins in breast milk in the same composition was 5.0, 0.10, 0.1, 0.1, and 0.1 ppb, respectively [70].
The concentration of DDT insecticide in the muscle tissue of whitefish on the SSCS was further investigated and it was concluded that along the SSCS, the average concentration of DDT toxin was 0.0035 parts per million in adipose tissue. Among the 5 stations, the samples taken from Fereydoonkanaar and Bandar-e-Turkmen stations (with an average of less than 0.0003 parts per million in adipose tissue) had the lowest rate of DDT toxin, while samples gathered from Chalous station (with 0.016 parts per million in adipose tissue) recorded the highest DDT content. In general, in the SSCS, the amount of the toxin is less than the allowable daily absorption, yet due to the high toxicity and stability of this compound, its amount in species such as white fish, which have higher economic value, should be minimized [71].
Toxicology of organophosphates in humans
Bioconcentration which is a known process for food chain in terms of residual toxins and humans, is always at the top of a food chain, so maximum chance for accumulation of pesticides in the human body is always inevitable. Humans can properly absorb organophosphates through a variety of pathways, including inhalation, ingestion, and skin penetration. Therefore, the relative uptake of this organophosphate is directly affected which result in a significant difference in relative uptake through these different pathways. Such organophosphate compounds can also directly affect humans, various species of birds, amphibians, and mammals [72,73].
The use of OP pesticides and CM pesticides make up about 70% of the total pesticide application, so it is required to get ensured that pesticides present in fruits and vegetables are destroyed before their sale. In this regard, performing a rapid, sensitive, and cost-effective analysis of pesticide residues is quite essential to ensure the quality of agricultural products [9].
Organophosphates have a high tendency to phosphorylate the AChE and to reduce AChE availability. The AChE is primarily required for normal control of neurotransmission from nerve fibers to smooth and skeletal muscle cells, secretory cells, and autonomic ganglia as well as in the central nervous system (CNS). Due to shortage of available AChE, the amount of shock transporter, acetylcholine, is produced to over- stimulate human organs at nerve endings. AChE phosphorylation can inactivate the acute percentage of tissue enzyme mass, so that based on these signs, the symptoms of cholinergic poisoning are recognized. A proper dose of organophosphate compounds cause enzyme function considerable loss of enzyme function, and AChE solution in cholinergic-effective neuronal connections (muscarinic effects), skeletal nerve muscle connections, and autonomic ganglia (nicotine effects). These cholinergic neural connections consist of different smooth and secretory muscle cells and are active in accumulation of high concentrations of AChE in these connections, which are the result of muscle contraction and secretion. On the other hand, in case of skeletal muscle connections, it causes muscle strain and respiratory paralysis. However, high concentrations of AChE cause sensory and behavioral disorders, incoordination, depressed motor function, and respiratory depression in CNS. Increased pulmonary secretions along with respiratory failure are common causes of death by organophosphate poisoning. Therefore, final improvement depends on production of new enzymes in such vital sites [1,3,19].
To visually determine the residual pesticides and to improcve accuracy, it is essential to consider any increase in color resolution. The results of research on visual acuity also revealed that human visual cells are sensitive to wavelengths that vary adjacent to water temperature [74]. Thus, indoxy acetate is proposed as a chromogenic reagent [9].
Indoxyl acetate is a good chromogenic substrate of acE which is used for determining enzyme activity. Hydrolysis of indoxyl acetate is also decomposed by using AChE to produce indoxyl (Figure 4). Indoxyl is then rapidly oxidized to form indigo, which is completely green-blue and can be detected by the naked eye; thus, due to the change in color intensity, which is caused by inhibition of acE activity, the residual concentration of pesticides can be easily analyzed by qualitative or semi-quantitative methods. As the screening card provided is provided, a more sensitive candidate for rapid detection of pesticide residues in real samples will result [9].
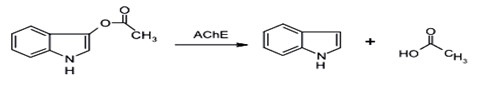
Figure 4: Reaction mechanism of indoxyl acetate hydrolysis reaction catalyzed by AChE
Advances in extraction and purification methods of residual pesticides from food matrix
Each analytical method is composed of several specific methods including sample preparation, separation and division of optimal analysis, concentration analysis and data, respectively [75]. In the case of food and environmental samples, separation of pesticides from residual matrices is a rather dynamic phenomenon for accurate analysis and detection, even at very low concentrations [76]. During the separation of the residual components with some chemicals taken from the plant food matrix, proteins, tannins, lipids, waxes, chlorophyll, and terpenoids are further extracted along with the corresponding solvents, which tend to inhibit the reaction of pesticides with chromogenic reactions. Moreover, pigments and dyes are directly involved in analysis of colorimetric analysis, and in turn, can contaminate columns and detectors during analysis. Accordingly, to solve this problem, the methods of isolation, division, and purification play a crucial role in sample preparation are used [77]. From this perspective, numerous researchers have reported significant aspects of separation and fractionation techniques such as SFE, SPE, SPME, MAE, ASE, MSPE, DLLME, which are quick, easy, cheap, effective, robust and safe (QuEChERS) [78]. Subsequently, researchers have updated and reported an abundance of purification processes including steam distillation, simultaneous vacuum distillation, coagulation, soaping, oxidation, adsorption, and chromatography [75].
Nowadays, the QuEChERS technique is increasingly getting popular, and based on this technique, several methods have been published to identify and analyze organic phosphorus pesticides in food and environmental samples. The QuEChERS technique is mainly used as an unusual method for distribution of persistent components of pesticides in vegetables and fruits. SPE with desalination effect and dispersed SPE, which are used for sample purification, are the most pervasive methods for this technique. Both steps are significantly used to identify and analyze a large set of pesticides from different food matrices, but combining these techniques has led to a great revolution in analysis of residual pesticides [79].
Since high recovery of residual toxins is always desirable during analysis and identification, several modifications for the proposed QuEChERS method [80] mainly involve applying different combinations. The first type of modification focuses on pH adjustment done by using acetate and citrate buffer to prevent degradation of pesticide-sensitive pesticides. For this purpose, buffers such as acetate buffers, citrate buffers, formats, and phosphate buffer salt are used for stability of pesticides [81].
Various mixtures of solvents such as acetonitrile: methanol, acetonitrile: hexane, methanol: water are used for complete extraction of pesticides from complex food matrices [82]. Decreased ionization of residual pesticides occurs mostly during LLE extraction, so to overcome this problem, N-propylethylendiamine is used in combination with C18 and ethylene diamine propyl is also used in combination with graphitized carbon black [81]. Besides, in presence or absence of N-propylethylendiamine, integration of C18 with graphite black carbon is reflected as an important component in the advanced QuEChERS method. In this regard, various salts such as NaCl and MgSO4 are used in this method to remove excess water in a form bound to pesticides. Also, to prevent exothermic hydration reactions, QuEChERS method was used at low temperatures or with freezing process [80].
Extraction solutions, extraction methods, and purification methods for mycotoxins
Mycotoxins are the most widespread biological toxins that even in very low concentrations, can contaminate foods. It is proved that some emerging extraction techniques and current analytical techniques and methods are effective both in detection of mycotoxins and in identification of mycotoxin-producing fungi. Chromatographic techniques such as HPLC with various detectors such as fluorescence, diode array, UV, LC with MS, and in situ LC - MS are powerful diagnostic and analytical tools. Recent advances are also introduced in development of rapid safety-based diagnostic techniques such as immunoassays and biosensors, as well as emerging technologies such as proteomic and genomic methods, molecular techniques, electronic nose, dye diffusion, quantitative NMR, and spectroscopic imaging, which are used to detect mycotoxins in food [34].
In extraction of edible and contaminated samples which is suitable for removing mycotoxins from the sample, appropriate solvents are used. The choice of solvents and extraction method plays a significant role in successful extraction. An optimal extraction solvent is the one that just removes mycotoxins with the highest efficiency from the sample. Methanol-water and acetonitrile-water mixtures in different proportions have been frequently used in extraction and analysis of mycotoxins [83]. Pigments, essential oils, and fatty acids present in the samples make extraction difficult, so they require using various extraction solvents such as a mixture of ethyl acid-formic acid [84]. Moreover, other extraction solvents such as 1-octanol and toluene [85], dichloromethane [86], acetone [87], and chloroform [88] are used to extract mycotoxins. Although hydrophobic mycotoxins can be easily dissolved in all previous solvents, polar mycotoxins such as FB are soluble in water [88]. Deep eutectic solvent (DES), recently emerged as a new green solvent [89], has successfully extracted aflatoxins from rice samples. This extractant is made of a combination of two components (tetramethyl ammonium chloride and malonic acid) with biodegradable, safe, and economical capability.
Diagnostic methods based on immunochemical assays
Fungal toxins or mycotoxins are light weight compounds produced by some saprophytic fungi as secondary metabolites and are very harmful to both humans and domestic animals when exposed to livestock or plants. These toxins have a more complex chemical structure than bacterial toxins. So far, 80- 90 types of mycotoxins secreted by about 120 species of fungi are known in nature. Toxin-producing fungi include Aspergillus, Fusarium, Penicillium, Alternaria, and Clavisps, among which the fungal toxins aflatoxins and ochratoxins are more dangerous with detrimental effects on farmed animals like fish among which one can refer to reduced body immunity loss of growth and production, increased feed conversion ratio, and high aquatic losses [90].
There are also reports on the negative effect of this toxin on the immune system, the development of liver diseases, the effects of malignancy, and carcinogenesis [91]. On the other hand, it causes degenerative changes in liver parenchyma cells, including nuclear and cytoplasmic edema and necrosis of proximal renal tubules [92].
According to the report of the WHO Joint Committee and the Food and Agriculture Organization (FAO), each person can tolerate a maximum of 14 ng/ kg of body weight of ochratoxin A per day, and greater doses may lead to kidney disease [88].
Rapid detection methods are often based on immunochemical assays. Some major instances are ELISA, dipsticks, flow membranes, and LFD. Among all immunological methods, ELISA is the most important tool for the rapid detection and quantification of mycotoxins [88]. This technology was developed on interactions between antigen-antibody complex and chromogenic substrates, and the measurable result was obtained from color spectrophotometric measurements [93]. ELISA methods were also developed for AF, ZEA, OTA, DON, T2 / HT2, and FB tests in various agricultural commodities [94,95].
In Dipstick, the operational principle resembles ELISA's, so a membrane-based immunoassay method provides qualitative and semi-quantitative results. Despite the high accuracy of the results, all of these methods have poor commercial performance safety [88], yet they are fast and the given results can be used within minutes.
LFDs, also called safety strips or safety tapes, which are fast have in-situ screening tools for immunochromatographic tests that work in a competitive manner. They also have antibodies labeled as signal reagents [96], which are used as pregnancy tests. The results of these tests, either positive or negative, are obtained from the visual evaluation. Moreover, photometric stripe readers can be used to obtain the required result [93]. Signal amplification in LFD is achieved through new materials such as quantum dots (QD) [97], AuNPs [98], magnetic nanoparticles (Fe3O4) [99], and carbon nanoparticles (CNP) [100]. Although this method has great advantages, a major reason for its limited use is its interference. In addition, it is a complex matrix for identifying tracking analyzes [83]. Also, its limited application is associated with reproducibility, reliability with different matrices, and sensitivity [101].
Conclusion
Today, pesticides are mainly used to improve cultivars with higher yields. Pesticides are frequently used to protect plants against harmful organisms and pests such as insects, weeds, rodents, or fungi. However, due to their inherent toxicity, their application can in turn lead to long-term harms to the environment and biodiversity. Some pesticide metabolites are in contact with the environment, whereas others remain as residues. For example, Chlorine pesticides are resistant to environmental degradation caused by chemical, biological, and photolytic processes, so their bioaccumulation in tissues and food chains will harm human and environmental health. In addition, undigested pesticide residues accumulate in the organs and can affect human health in the long run. Therefore, it requires ongoing monitoring of pesticides remaining in food and the environment to ensure comprehensive compliance with legal restrictions.
Funding
Not applicable.
Authors' contributions
All authors had equal role in study design, work, statistical analysis and manuscript writing.
Consent for publications
All authors approved the final manuscript for publication.
Conflict of Interest
The authors have no conflict of interest to declare.
Availability of data and material
Data are available on request from the authors.
Ethics approval and consent to participate
No human or animals were used in the present research.
HOW TO CITE THIS ARTICLE
Bahman Fazeli-Nasab, Laleh Shahraki-Mojahed, Zahra Beigomi, Maryam Beigomi, Anahita Pahlavan. Rapid detection Methods of Pesticides Residues in Vegetable Foods, Chem. Methodol., 2022, 6(1) 24-40
DOI: 10.22034/chemm.2022.1.3