Document Type : Original Article
Authors
Department of Chemistry, College of Education for Pure Science (Ibn Al-Haitham), University of Baghdad, Adhamiyah, Baghdad, Iraq
Abstract
A new Azo-Schiff base ligand was prepared by reaction of 2-aminobenzothiazole and 2-hydroxy-1-naphthlaldehyde to form the azo compound. In the latest step, the azo compound was reacted with thiosemicarbazide to give the ligand. The synthesized ligand was used for complexes with different metal ions like Ni(II), Co(II), Cr(III), Mn(II), and Cu(II) by using a molar ratio of ligand: metal as 1:2. Resulted in compounds were characterized by FT-IR, 1HNMR, 13CNMR, UV–Vis spectroscopy, Mass, elemental analysis, magnetic moment and molar conductivity studies. The ligand and its complexes’ activity were tested against four types of bacteria and one type of fungi; some compounds gave a positive result, and others showed negative inhibition.
Graphical Abstract
Keywords
Main Subjects
Introduction
The presence of the azo moiety distinguishes the azo compounds or dyes (–N=N–) in their structure, conjugated with two distinct or identical mono- or polycyclic aromatic or hetero-aromatic systems [1]. Azo dyes are synthesized via two steps reaction, the first being the synthesis of an aromatic diazonium ion from an aniline derivative. The second step is coupling the diazonium salt with an aromatic compound [2]. The azo groups generally connected to benzene and naphthalene rings but can also be attached to aromatic heterocycles or enoliz able aliphatic groups [3]. Azo dyes acquired a broad interest in application to a biological system and as indicators in complex colorimetric titration in analytical chemistry [4]. The Schiff base reaction refers to the interaction between compounds having amino groups (NH2, NH2OH, etc.) and carbonyl groups (aldehydes or ketones); Schiff bases are compounds that include an azo methine group (-CH=N-) [5]. In 2021, a new azo-Schiff base ligand was prepared by coupling reaction of the diazonium salt with an appropriate amount of (imidazole derivative) as a coupling component in an alkaline solution [6]. The study aimed to synthesize and characterize new azo-Schiff compounds and its metal complexes, then test the ligand and complexes’ efficient biological activity against various types of bacteria and fungi.
Materials and Methods
All material and chemicals obtained from Aldrich chemical company (USA) and Alfa Aesar and athier. The ligands NMR spectra 1H-NMR and 13C-NMR were evaluated in DMSO-d6 with a Brucker 300 MHZ instrument (300 MHZ for 1H-NMR and 125 M HZ for 13C-NMR), using tetramethyl silane TMS as an internal reference. The FT-IR spectra as KBr discs were recorded using FT-IR-600 Fourier transform infrared spectra in the range 4000-400 cm-1. The electronic spectra of compounds were determined using a (UV-Vis) spectrophotometer type Shimadzu UV-160 in the range 1000-200 nm. The samples were solvated in DMSO using a 1.0 cm length cuvette cell with a concentration of 10-3 mol. L-1 at room temperature. Mass spectra were obtained by (+) electrospray technique using Agilent mass spectrometer Sciex ESI MS. Melting points of prepared compounds were recorded using an electrothermal Stuart apparatus, model SMP 40 US. Magnetic moments at 308 K were measured with a magnetic susceptibility balance on Sherwood Scientific Devised. The complexes’ molar conductivities were measured at room temperature using Eutech instruments cyberscan con (510) digital conductivity meter for (10-3 10-5 M solutions) of the compounds in Dimethyl sulfoxide solution (DMSO). Compounds were tested against four different bacteria and one species of fungi.
Synthesis of HL
Synthesis of thiosemicarbazide ligand [2-((4-(benzo[d] thiazol-2-yldiazenyl)-3 hydroxynaphthalen-2-yl)methylene)] hydrazine carbothioamide] from azo derivative [(4-(benzo [d]thiazol-2-yldiazenyl)-3-hydroxy-2-naphthaldehyde)] which synthesized by reaction of 2-amino benzothiazole (1.5 g, 9.9 mmol) and sodium nitrite (0.689 g, 9.9 mmol) combined in 10 mL of water, chilled to 0-5 °C and then mixed with hydrochloric acid with vigorous stirring to prepare azo compound according to the literature [7]. The cooled solution of 2- hydroxyl-1-naphthaldehyde (1.7 g, 9.9 mmol) and NaOH (0.399 g, 9.9 mmol) was then added to the diazonium salt solution, and the precipitate formed at pH=4 was filtered and rinsed many times with cold water before being dried at pH 6-7. The orange precipitate was filtered out, yielding (2.221 g, 66.71%) with MP 79-81 °C, after thiosemicarbazide solution was added (0.274, 2.9 mm) to azo derivative (1 g, 2.9 mm) with a few drops of glacial acetic acid as a catalyst, refluxed 6 hrs, then left at room temperature extended to 15 minutes. The precipitate compound was obtained by filtration, washed with ethanol, and dried; light yellow product was collected. Yield: 0.894 g, 73.33 % M.P>300 C. The synthesis of the ligand [HL] is illustrated in Scheme 1.
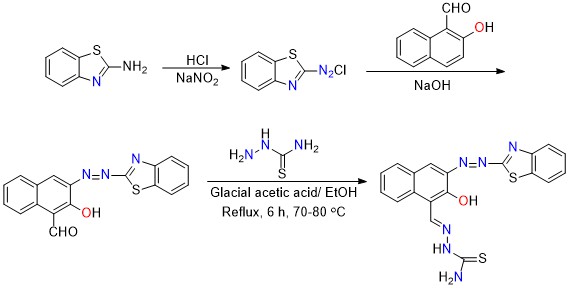
Scheme 1: Synthesis route for HL1
Synthesis of complexes
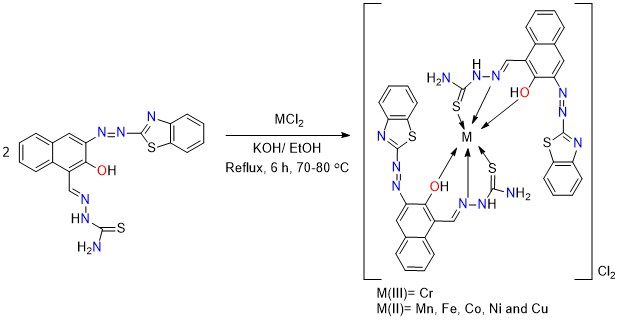
The complexes were prepared by dissolving of HL ligand (0.4 g, 0.49 mmol) in 18 mL ethanol, followed by adding an equivalent amount from KOH (0.168 g, 0.49 mmol), followed by adding the metal salt CoCl2 (0.0638 g, 0.49 mmol) as dropwise on the mixture of the ligand; the mixture was refluxed for 6 hrs with continuous stirring at fixed temperature 70-80 °C. The mixture was filtered, and the resulted participate was washed several times with cold ethanol; other complexes were prepared using the same method. Scheme 2 shows the Synthesis route for metal complexes. Melting point of Complexes, colors and yields are all included in Table 1.
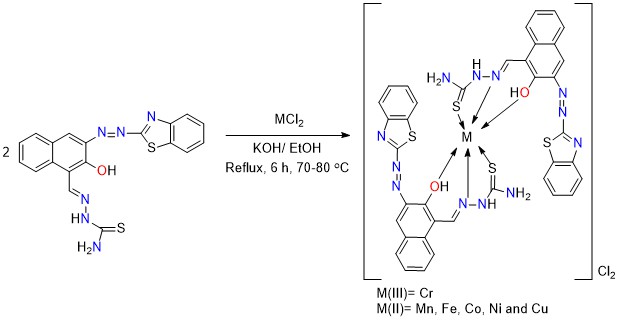
Scheme 2: Synthesis route of metal complexes
Table 1: Metal salts quantities, colours, yields and melting points of complexes of HL1
|
Complex of Metal ions |
Weight of metal salt (g) |
Complex weight (g) |
Color |
mp (°C) |
Yield (%) |
|
[Cr(HL)2]Cl3 |
0.14 |
971.32 |
Yellow |
> 300 |
81 |
|
[Mn(HL)2]Cl2 |
0.10 |
938.81 |
light brown |
> 300 |
75 |
|
[Co(HL)2]Cl2 |
0.07 |
942.80 |
Dark Brown |
> 300 |
85 |
|
[Ni(HL)2]Cl2 |
0.12 |
942.56 |
Brown |
> 300 |
77 |
|
[Cu(HL)2]Cl2 |
0.09 |
947.41 |
Green |
> 300 |
81 |
Results and Discussion
All complexes were synthesized by reacting the metal salts with the Schiff base in a 1:2 mole ratio (M: L). All of the complexes were colored solid compounds. Table 1 shows that the analytical findings corresponded with the proposed formula of a 1:2 M: L molar ratio. The complexes synthesized using HL were soluble in dimethyl form amide and dimethyl sulfoxide, which has partial solubility in ethanol, methanol, benzene, and water. Some of the physical and chemical characteristics are shown in Table 2.
Table 2: Elemental microanalysis data and some physical properties for complexes

FT-IR spectrum
Table 3 displays the spectrum of precursor (azo); the band at 3159 cm–1 is referred to as the phenolic OH group. Bands at 3074 cm-1 and 2889 cm-1 referred to u(C–H) aro and u(C–H) ald groups respectively [8], the bands at 1643, 1627, (1608), 1465 and 1315 cm-1 referred to C=N, C=O, C=C, N=N and C=S groups respectively. Also, in Table 3, numerical values of Schiff base ligand showed a band at 3448 cm-1 and 3375 cm-1 that refers to OH and NH groups, respectively, in HL, and 3263 and 3167 cm-1 referred to NH2 attributed to asymmetrical and symmetrical stretching bending respectively. Also, bands appeared at 3051 and 2978-2908 cm-1 that were assigned to v(C–H)aro and v (C–H)ımıc, the bands at 1624, 1608, 1465, and 1361 may be assigned to C=N, C=C, N=N, and C=S groups respectively [8, 9]. The spectrum of the complexes showed shifting in the imine band, phenolic band, and thione band, the shifting range was 1620-1612 cm-1 in the imine [9], and the C-O shifting range was 3468-3429 cm-1 [9, 10], while the thione band from 1361-1300. The azo group did not show any noticeable shift, which means the group did not participate in the coordination with the metal ions. Also, new bands for stretching frequencies of M-N have been allocated at 596-570 cm-1, and another band was observed at range 497-470 cm-1 indicating the formation of M-O coordination bond and range 455-420 cm-1 ascribed M-S bond [8-10].
1H and 13C -NMR spectra of the ligands
According to the literature review, the NMR spectroscopy was required to determine the structure of various types of compounds; NMR spectra were recorded in DMSO-d6 (dimethyl sulfoxide) using TMS (tetramethyl silane) as standard.
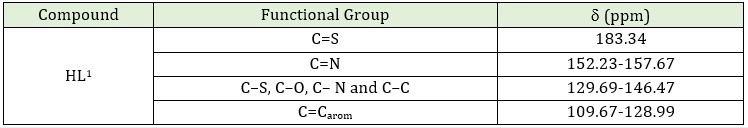
The 1H-NMR spectrum of HL is shown in Table 4; the singlet signal of the phenolic proton OH appeared at 11.42 ppm. The small signal also appears as a singlet in 10.52 ppm may be assigned for NH, and the signal at 9.04 ppm may be attributed to the NH2 group [8]. The spectrum of Schiffʼs base showed a peak at 8.55 ppm, which indicates the formation of the azomethine group (-CH=N) [11]. The multiple chemical shifts around 7.19-8.55 ppm may be assigned to aromatic protons [12]. The 13C-NMR spectrum of HL, Table 5 in DMSO–d6 solvent shows a chemical shift at the range 129.69-146.47 ppm can be attributed to C-S, C-O, C-N and C-C groups [9]. The chemical shift at 152.23 is attributed to azomethine endo, while 157.67 ppm can be attributed to azomethine group (C=N) [10]. The chemical shift in the range 109.67-128.99 could be attributed to the C aromatic group [9]. Finally, the chemical shift at 183.34 ppm can be attributed to the C=S group [8, 10, 11].
Table 3: The FT-IR spectral data of compounds (cm-1)
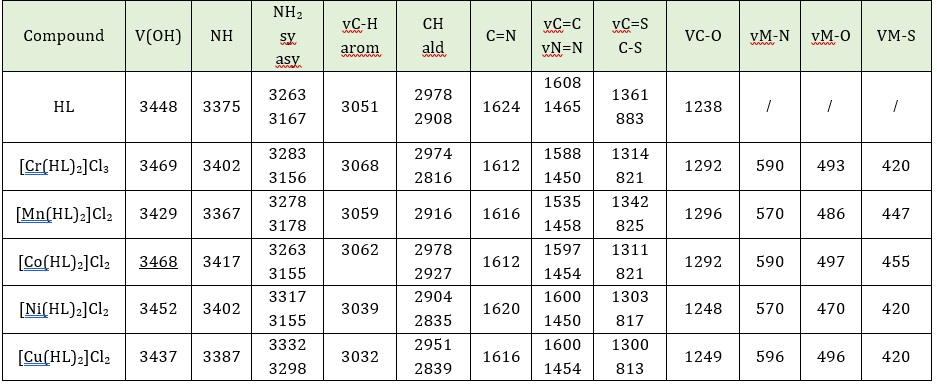
Table 4: 1H-NMR spectral data for the ligand HL1
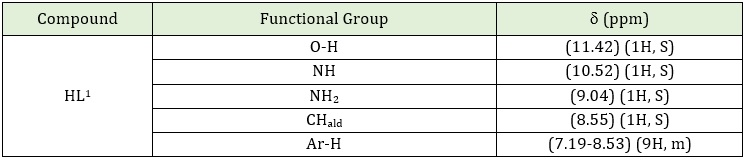
Table 5: 13C-NMR spectral data for the ligand HL1
Mass spectroscopy
Table 6 shows the Schiff base ligand’s mass spectrum, which has a well–defined molecular ion peak at m/z = 406.484 amu, which correlates (M+1) with the Schiff base ligand's molecular formula (C19H14N6OS2). The spectra of the HL ligand reveal a succession of peaks at m/z, 380.46, 264.33, 174.20, and 120.15, amu, which correspond to the fragments. The strength of these peaks indicates the pieces’ stabilities [13].
UV-Vis and the magnetic susceptibility measurement
In Table 7, the ligands’ absorption spectra revealed peaks at 265, 329, and 381 nm due to the π→π* and 345 related to n→π* transition [2]. The range of peaks at 271-387 nm may be related to intra ligand for complexes 1-5 [14], while the range of peaks at 416-438 nm revealed to (CT) for complexes [8-10]. Finally, the peaks in the visible region can be associated with d-d transitions. The electronic spectrum of Co(II) displays the peaks at 461 and 803 nm related to4T1g(F) →4T1g(P) and 4T1g(F) →4T1g(F) transitions, respectively, octahedral geometry around the central metal ion was suggested [14]. The electronic spectrum of Cu(II) complex displays peaks at 429 nm may be revealed to 2B1g→ 2Eg transition, and at 760-799 related to 2B1g→2A2g transition, which revealed that the complex own octahedral geometry [14, 15]. The electronic spectrum of Mn(II) displays the peaks at 734, 780, and 821 nm related to 6A1g→4T1g and 6A1g(s)→4T1g(G) transition, the octahedral geometry around the complex was suggested [16, 17]. The electronic spectrum of Cr(III) displays the peaks at 679 and 848 nm related to 4A2g(F) →4T2g (F) [7, 8], and octahedral geometry was proposed [9]. The electronic spectrum of the Ni(II) complex displays the peak at 855 nm related to 3A2g(F)→3T1g (p), which suggests an octahedral geometry around the central metal ion [14]. The magnetic moment of the complexes Ni(II), Cu(II), Co(II), Mn(II), and Cr(III) value agrees with the electronic spectrum transition and the suggested structures.
Biological activities
The effect of the synthesized compounds was evaluated using the agar diffusion technique on four types of bacteria (E. Coli, Enterobacter S aureus, and B. Subtilis) and one type of fungi (Candida) (Table 8). The inhibition zone (IZ) of 1×10-3 M of the compounds was compared to the IZ of Ceftriaxone (BP) antibiotic as standard [18, 19]. The solvent was conducted, and its effect on bacterial growth –ve (zero) [20]. The bacteria and fungi were chosen for their recognized impact on various illnesses; they exhibit varying resistance to medicines and medicinal compounds. The ligand and its complexes demonstrated a reasonable zone of inhibition.
Table 6: The mass fragmentation data of HL1
|
No. |
Fragment |
Mass/Charge (m/z+) |
|
1 |
C19H14N6O S2 |
406.48 |
|
2 |
C18H14N5OS2+ [M+1] |
380.46 |
|
3 |
C15H10N3S+ |
264.33 |
|
4 |
C 8H4N3S+ |
174.20 |
|
5 |
C6H2NS- |
120.15 |
Table 7: Electronic spectral data of the ligand and ıts complexes

Table 8: The assessment of ligands and their complexes from biological perspective

Conclusion
It is reported that the synthesis and characterization of azo Schiff base ligand by reacting (4-(benzo [d] thiazol-2-yldiazenyl)-3-hydroxy-2-naphthaldehyde) with thiosemicarbazides to produce HL. The ligand interaction with metal salts to produce complexes more stable the imine group atoms (N), (OH), and (C=S) performed as tridentate coordinates. Uses such as FT-IR, UV-Visible, 13C, 1H-NMR, molar conductivity, and mass, magnetic, susceptibility indicated that all complexes were produced to adopt an octahedral geometric form. When tested for biological effectiveness against four types of bacteria (E. Coli, Enterobacter S aureus, and B. Subtilis) and one type of fungi (Candida), all of the synthesized compounds showed adequate inhibition in various ranges against the bacterium spices; also, they show the effect on the fungi spices.
Acknowledgements
The authors extend sincere thanks to Department of Chemistry, College of Education for Pure Science (Ibn Al-Haitham).
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
Conflict of Interest
There are no conflicts of interest in this study.
ORCID:
Khadija F. Mohammed
https://orcid.org/0000-0003-4449-1764
HOW TO CITE THIS ARTICLE
Khadija F. Mohammed, Hasan A. Hasan. Synthesis, Chemical and Biological Activity Studies of Azo-Schiff Base Ligand and Its Metal Complexess. Chem. Methodol., 2022, 6(12) 905-913