Document Type : Original Article
Authors
1 Department of Chemistry, Faculty of Science, Ilam University, Ilam 69315516, Iran
2 Department of Chemistry, University of Zanjan, Zanjan, 45195-313, Iran
Abstract
The primary objective of this study was to devise a straightforward and environmentally sustainable approach for creating Biginelli compounds. Accordingly, a p(AMPS) hydrogel served as an effective catalyst of diverse nature, facilitating the preparation of 3,4-dihydropyrimidine-2-(1H)-ones, commonly recognized as Biginelli compounds. The chemical process unfolded using an eco-friendly method, resulting in notably abundant product yields. Moreover, the catalyst exhibited remarkable reusability, maintaining its efficacy through four cycles without a substantial decline. The innovative Biginelli reaction technique achieves environmentally conscious synthesis, robust product generation, simplified isolation procedures, catalyst sustainability, and avoids harmful by-products.
Graphical Abstract
Keywords
Main Subjects
Introduction
Hydrogels, being three-dimensional hydrophilic polymer networks, offer distinct environments suitable for preparing and safeguarding metal nanoparticles. Their three-dimensional, water-swollen, and cross-linked structure of hydrophilic polymer chains makes them particularly ideal for this purpose. Polymer chains within hydrogel networks have hydrophilic properties that are caused by the functional groups SO3H, -COOH, -CONH2, -OH, and -NH2 present in them. Recently, hydrogels have received attention as an intriguing class of catalysts, motivated by the rising need to develop cost-effective, environmentally friendly, efficient, and reusable catalysts [1-3]. Green chemistry stands out as an efficient technology aimed at minimizing or preferably eliminating waste generation, avoiding toxic solvents and reagents, and maximizing the use of renewable raw materials whenever possible [4-9].
Multi-component reactions (MCRs) are highly efficient and versatile synthetic procedures that promote the production of unique and complex molecular structures. They offer advantages over typical multistep syntheses [10-15]. In the realm of organic synthesis, multicomponent reactions (MCRs) that specifically target the synthesis of heterocyclic molecules featuring the dihydropyrimidinone scaffold have earned considerable acclaim [16-20]. The recent surge of interest in exploiting the MCRs capabilities underscores their pivotal role in advancing innovative synthetic methodologies [21-23]. Among various MCRs, the Biginelli reaction holds paramount importance. This chemical pathway enables the production of dihydropyrimidinones (DHPMs), a technique initially published by the Italian chemist Pietro Biginelli over a century ago [24-28].
The use of environmentally friendly methods to produce multi-component compounds is widely favored. Sustainable techniques reduce the environmental impact of chemical processes [29-31]. Green synthesis uses non-toxic solvents, sustainable resources, and energy-saving techniques [32-35]. Researchers can improve chemical production by prioritizing these principles and developing synthetic routes that produce compounds. The synthesis of this multi-component green compound contributes to global efforts to reduce the environmental impact of chemical production [36-40].
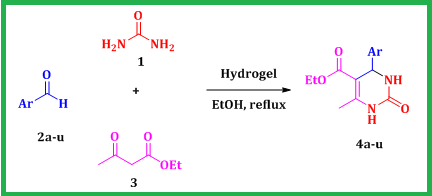
Despite their potential advantages, several of the documented one-pot protocols have limitations, including the utilization of costly reagents, reliance on volatile strong acid conditions, and long reaction times. Consequently, there is a pressing need to introduce a more moderate and efficient method that not only avoids these drawbacks, but also ensures higher yields. To the best of our knowledge, there are no documented instances employing hydrogels as eco-friendly additive within a green solvent under reflux conditions. Consequently, this study introduces a novel methodology for synthesizing substituted DHPM, employing a straightforward and suitable approach under green conditions, as exhibited in Scheme 1.
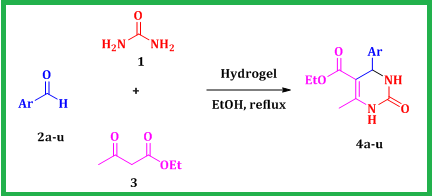
Scheme 1: DHPMs synthesis using hydrogel
Materials and Methods
Merck, Fluka, and Aldrich supplied starting materials used without purification.
Preparation of Catalytic Hydrogel
The process detailed in [41] was followed to create Poly (2-acrylamido-2-methyl-1-propanesulfonic acid) (p(AMPS)) hydrogel. This involved radical polymerization using a redox initiator. Specifically, 0.0217 mol of AMPS (C7H13NO4S) was mixed with 0.5 mol% of Bis (C7H10N2O2) in 3.5 mL water along with 100 mL TEMED (C6H16N2). A 1 mL aqueous initiator solution (5 wt%) of APS ( (NH4)2S2O8) (1 mol% in relation to the monomer) was added to the hydrogel precursors.
The mixture was then poured into plastic pipettes with a 4 mm diameter and allowed to undergo polymerization and crosslinking for 24 h at room temperature. The resulting p(AMPS) hydrogel was cut into cylindrical shapes measuring 4-5 mm in length. These pieces were cleaned by immersing them in water for three days, with water changed per 12 h to remove residues like monomer, polymer, crosslinker, and unreacted initiator. Post-cleaning, the hydrogel was dried in an oven at 45 °C until reaching a constant weight. Finally, it was stored in an airtight container for future use.
General Method for the Synthesis of DHPMs Derivatives
A mixture of urea (2 mmol), aldehyde 2a-u (1 mmol) and hydrogel (0.15 mg) in ethanol (5 mL) was subjected to magnetic stirring at room temperature. After adding 1.5 mmol of ethyl acetoacetate, the mixture was stirred under reflux conditions for a specified time until the reaction was completed.
The reaction was monitored by TLC (n-hexane:EtOAc, 10:6). At the end of the reaction, the solid product was filtered and purified through recrystallization in hot ethanol. All identified products, recognized as known compounds, underwent characterization including analysis of melting points, IR, 1H-NMR, and 13C-NMR spectra.
The Selected Spectral Data
Sample 4b: mp 212-214 °C; IR: NH 3231, NH 3102, CO 1701, CO 1646, C–O 1122, and C–N 1091 cm-1; 1H-NMR (250.13 MHz, CDCl3): δ 1.04 (t, 3J 7.5 Hz, 3H, CH3), 2.41 (s, 3H, CH3), 3.98 (q, 3J 7.5 Hz, 2H, CH2), 5.86 (s, 1H, CH), 7.18–7.37 (m, 4H, Ar–H), 5.86 (NH), and 8.92 (NH); 13C-NMR (62.90 MHz, CDCl3) δ 13.9, 18.3, 52.1, 59.9, 98.8, 127.5, 128.0, 129.8, 132.5, 139.5, 148.5, 153.3, and 165.3 ppm.
Results and Discussion
A practical and straightforward method was employed in this study to affirm the synthesis of DHPM. Various reaction setups were explored to establish optimal conditions for creating modified DHPM (4a-u), involving urea, ethyl acetoacetate, 4-chlorobenzaldehyde, and hydrogel as an eco-friendly element in ethanol solvent, serving as a model reaction. In our efforts to optimize these reaction conditions, we investigated the influence of solvents and the amount of additives. We initially experimented with different solvents include: H2O, C2H5OH, C2H5OH-H2O, CH3OH, CH3CN, and CH2Cl2 in the synthesis of 4c, a representative compound. The most favorable result was obtained from the reaction using 4-chlorobenzaldehyde, ethyl acetoacetate, urea, and hydrogel as a green additive in ethanol solvent, which provided 4c in 6 h with 94% yield (Table 1, Entry 2).
While H2O, C2H5OH-H2O, CH3OH, CH2Cl2, and CH3CN resulted in moderate yields of the desired products, C2H5OH proved to be the optimal solvent for all subsequent processes.
To determine the optimized quantity of the catalyst, the model reaction was conducted for the synthesis of compound 4c. The additive quantity was varied, as depicted in Table 2, entries 2-4. The highest yield of the desired product was attained with 0.15 mg of the additive. The summarized results are presented in Table 2. Under these optimized conditions, we extended our examination to the reaction of ethyl acetoacetate and urea with diverse aldehydes using hydrogel as an efficient and thermally stable catalyst.
Table 1: The effect of the solvents on the preparation of substituted 3,4-dihyrdopyrimidin-2-(1H)-ones
|
Yield a (%) |
Solvent |
Entry |
|
Trace |
H2O |
1 |
|
94 |
C2H5OH |
2 |
|
Trace |
H2O-C2H5OH |
3 |
|
78 |
CH3OH |
4 |
|
76 |
CH2Cl2 |
5 |
|
86 |
CH3CN |
6 |
aIsolated yield
The reactions progressed seamlessly in ethanol under reflux conditions, completing within 6 h. The results pertaining to the conversion of differently substituted aryl aldehydes into DHPM derivatives 4a–u are presented in Table 3.
The proposed mechanism for synthesizing substituted DHPM (4a-u) is depicted in Scheme 2. Given the established reflux chemistry of substituted DHPM, we propose an acid-catalysed condensation of urea (2) and an aromatic aldehyde (1) in EtOH. The hydrogel presence aids imine formation (4). Subsequently, ethyl acetoacetate (5) undergoes nucleophilic addition, generating an enol (6) that contributes to a 1, 3-dipolar intermediate. This intermediate reacts with the imine, followed by an intramolecular attack leading to the nitrogen-containing six-membered ring. Elimination of ethanol and hydrogel molecules yields the target product, as detailed in Scheme 2.
The catalyst's recyclability, reusability, and catalytic performance were evaluated. Post-filtration from the reaction mix, the catalyst underwent washing with diluted water, drying, and reuse in subsequent reactions multiple times. Remarkably, the hydrogel exhibited consistent reusability for at least four cycles, maintaining product yield without noticeable decline. The potential for reusability and recycling of the hydrogel was explored via sequential condensations involving ethyl acetoacetate and 4-chlorobenzaldehyde with urea. Recovering the catalyst involved washing the reaction mix with water. The catalyst extracts, obtained after water evaporation and concentration, were reused without purification in the subsequent run. Impressively, the reaction proceeded smoothly, yielding products at 85-94%, highlighting the catalyst's sustained activity even after four runs (Table 4).
In summary, this innovative procedure offers an efficient route to synthesizing target products with high yields and exceptional purities. It represents a simple and convenient method applicable to various substituted DHPM. The structural confirmation of sample was validated through FT-IR, 1H-NMR, and 13C-NMR analyses.
Table 2: Influence of varying catalyst quantities on the synthesis of 4d
|
Yield (%) a |
Time (h) |
Catalyst (mg) |
Entry |
|
Trace |
10 |
None |
1 |
|
68 |
10 |
0.05 |
2 |
|
78 |
8 |
0.10 |
3 |
|
94 |
6 |
0.15 |
4 |
|
80 |
6 |
0.20 |
5 |
aIsolated yield
Table 3: Hydrogel Catalyzed synthesis of 3,4-dihyrdopyrimidin-2-(1H)-ones a,b
|
Entry |
Ar |
Product |
Yield |
Ref. |
|
1 |
Ph |
4a |
90 |
[42] |
|
2 |
4-Me-C6H4 |
4b |
86 |
[43] |
|
3 |
4-Cl-C6H4 |
4c |
94 |
[44] |
|
4 |
3-Cl-C6H4 |
4d |
88 |
[45] |
|
5 |
2-Cl-C6H4 |
4e |
86 |
[46] |
|
6 |
4-Br-C6H4 |
4f |
91 |
[47] |
|
7 |
3Br-C6H4 |
4g |
88 |
[48] |
|
7 |
4-NO2-C6H4 |
4h |
94 |
[49] |
|
8 |
3-NO2-C6H4 |
4i |
85 |
[50] |
|
9 |
4-OH-C6H4 |
4j |
84 |
[51] |
|
10 |
4-OMe-C6H4 |
4k |
89 |
[52] |
|
11 |
Thiophen |
4l |
85 |
[53] |
|
12 |
4-(Me)2N-C6H4 |
4m |
88 |
[54] |
|
13 |
Cinnamaldehyde |
4n |
86 |
[55] |
|
14 |
4F- C6H4 |
4o |
94 |
[48] |
|
15 |
3F- C6H4 |
4p |
89 |
[56] |
|
16 |
2F-C6H4 |
4q |
91 |
[57] |
|
17 |
Furfural |
4r |
84 |
[58] |
|
18 |
2,4-Cl-C6H4 |
4s |
92 |
[48] |
|
19 |
3MeO-4OH-C6H3 |
4t |
89 |
[48] |
|
20 |
4CN-C6H4 |
4u |
93 |
[56] |
aReaction conditions: Aldehyde 1 (1 mmol), urea 2 (2 mmol), ethyl acetoacetate 3 (1.5 mmol) EtOH (5 mL), and hydrogel (0.15 mg), Reflux condition. b Isolated yield
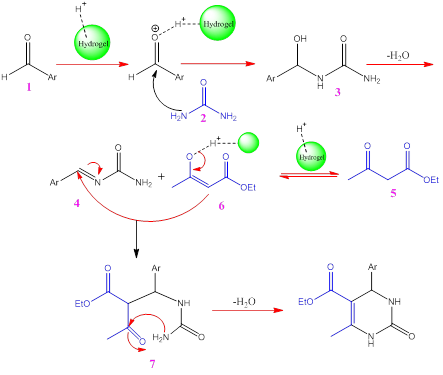
Scheme 2: The proposed mechanistic pathway for the DHPMs preparation
Table 4: Reusability and recovery of the catalyst
|
Yield (%)a |
Run |
|
94 |
Fresh |
|
94 |
First |
|
88 |
Second |
|
88 |
Third |
|
86 |
Fourth |
aIsolated yield
Conclusion
To sum up, a straightforward and effective method was introduced for producing a range of DHPMs. This method utilizes aryl aldehyde, β-ketoester, and urea as substrates, employing hydrogel as a catalyst. The approach boasts several benefits such as high yields, operational ease, straightforward product isolation via filtration, the use of an environmentally friendly solvent, and a significant reduction in yield after four reuses of the catalyst.
Disclosure statement
No potential conflict of interest was reported by the authors.
Funding
This study did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' contributions
All authors contributed toward data analysis, drafting, and revising the paper and agreed to responsible for all the aspects of this work.
Conflict of interest
The authors declare that they have no conflicts of interest in this article.
ORCID
Saeid Taghavi Fardood
https://www.orcid.org/0000-0002-0645-1393
Ali Ramazani
https://www.orcid.org/0000-0003-3072-7924
HOW TO CITE THIS ARTICLE
Saeid Taghavi Fardood, Zahra Hosseinzadeh, Ali Ramazani. Synthesis of 3,4-Dihydropyrimidin-2-(1H)-ones Using a Hydrogel as a Green and Reusable Catalyst. Chem. Methodol., 2024, 8(3) 154-163
OPEN ACCESS
©2024 The author(s). This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit: http://creativecommons.org/licenses/by/4.0/
PUBLISHER NOTE
Sami Publishing Company remains neutral concerning jurisdictional claims in published maps and institutional affiliations.
CURRENT PUBLISHER
Sami Publishing Company