Document Type : Original Article
Authors
College of Science Department of Chemistry, Baghdad University, Baghda, Iraq
Abstract
The determination of Cefotaxime sodium pure from and pharmaceuticals (tablets) by continuous flow injection scatter light has been created using a new, simple, rapid, and sensitive approach. Using a handmade ISNAG-FIuorimeter, the technique was predicated on the interaction of Cefotaxime sodium with vanadium oxide sulfate to produce a precipitate. The optimal value had a linear dynamic range of 0.01-20 mmoI/L for the instrument response to Cefotaxime sodium concentration, while the L.O.D was 0.334 g /sample from the stepwise dilution for the lowest concentration in the calibration graph's scatter plot range. For the scatter plot range and the linear dynamic range, the correlation coefficient (r) was 0.9769 and 0.9978, respectively, while percentage linearity (R2% percent) was 95.43 percent and 99.56 percent, respectively. With a concentration of 2.5 mmol/L, the RSD percent for repeatability (n=8) was lower than 0.23% percent for the determination of cefotaxime sodium. The proposed method was effectively used to determine the concentration of cefotaxime sodium in pharmaceutical tablets. The newly developed technique was compared with the conventional methods of analysis, i.e., Turbidemtric method and UV-spectrophotometer method, utilizing the usual addition method and paired t-test. The computed t-value at 95 percent confidence interval from the established approach revealed that there was no significant difference between the quoted figures of any individual firm.
Graphical Abstract
Keywords
Main Subjects
Introduction
Cefotaxime sodium is a sodium antibiotic (6R,7R) -3- [(acetyloxy)methyl] ((acetyloxy)methyl) ((acetyloxy)methyl) (( -7- (2Z) 2-(2- aminothiazol-4-yl)-2-(methoxyimino) acetyl] amino]-2-(2- aminothiazol-4-yl)-2-(methoxyimino) acetyl] amino] thia-oxo-5-thia-oxo-5-thia-oxo-5-thia-o-1-azabicyclo [4.2.0] C16H16N5NaO7S2, oct-2-ene-2-carboxylate]. It has a molecular weight of 477.447 and is a white or slightly yellow powder that is readily soluble in water but sparingly soluble in methanol. Figure 1 shows the chemical structure of CFTS [1].

Figure 1: chemical structure of CFTS
Cefotaxime is a broad-spectrum antibiotic that is typically used to treat bacterial infections of the skin, soft tissues, and urinary tract. It is a kind of antibiotic that is widely used. Cefotaxime sodium is a cephalosporin antibiotic that belongs to the third generation of cephalosporins. In addition to its antimicrobial action, third-generation Cephalosporins have a broad antibacterial range and resistance to beta-lactamase-producing organisms in addition to its antimicrobial activity, i.e., streptococci, staphylococci, pneumococci, etc. [2]. Cefotaxime failed to permeate the central nervous system and was unsuccessful in treating meningitis, but CFTS penetrates the central nervous system and achieves therapeutic concentrations, which are sufficient for treating meningitis caused by aerobic gram-negative bacteria [3]. It works against both Gram positive and Gram negative bacteria and has a broad spectrum of action. It is generally thought to be safe and effective in the same way as ceftriaxone is. These features are clinically and analytically significant. CFTS may be analyzed using a variety of analytical techniques that can be found in the literature. HPLC-UV detection, spectrophotometric Fluorescence, Electrochemical (Electrochemical-ISE, Electrochemical-MIP, turbidity using Ayah 4Sw-3D-T180o-2N90o-Solar-CFI analyzer [4], fluorescence resonance energy transducer, and continuous flow injection analysis via ISNAG-fluorimeter are some of these methods in their various forms and preparations [5]. In this research that was carried out with flow injection analysis with scattering light [6], measuring diverged beam of light at 0-90o angle was detected by homemade ISNAG- fluorimeter [7-10] via low-pressure mercury lamp as a source and using 2 sides [4 x 2.5cm] solar cells for the determination of CFTS with Vanadium oxide sulfate as a precipitating agent in the presence of Hydrochloride acid solutions.
Apparatus
A homemade ISANG fluorimeter was used with multichannel more than one-line feed. In this part of the research work only two lines, and four-channels peristaltic pump (Ismatec, Switzerland) and Six-port medium pressure injection valve were used. The output of measurement i.e; Ῡzi(mV)-tmin(dmm) was plotted by potentiometric recorder to determine the output signals (Siemens, Germany (1- 5 V). HANNA instrument for turbidity measurement was used similar to classical methods.
Materials and methods
Reagent and chemical
All chemicals were used of analytical-reagent and distilled water was used to prepare all the solutions. A standard solution of cefotaxime sodium (CFTS) (C16H16N5NaO7S2, molecular weight 477.447, 0.1mol.L-1) was prepared by dissolving 0.477447 g in 100 ml distilled water. We used a stock solution Vanadium oxide sulfat VOSO4, molecular weight 163.01 g/mol, 8.1505 g g/250 mL, Na2CO3 5.299 g /100 ml, CH3COONH4 3.884 g /100 mL, KCl 7.350 g/100 mL, NaCl 7.313 g/250 mL, NH4Cl 2.675 g/100 mL, CH3COOH 57.47 ml/L, HCl 88.28 ml/L and H2SO4 55.52 ml/L.
Sample preparation of Cefotaxime sodium (CFTS)
A batch of Twenty tablets were weighed then crushed and grinded; each of the drug containing 1000,500 mg of Cefotaxime sodium was weighed 0.48460 g, 0.40668 g and 0.40688 g (equivalent to 0.477447 g of active ingredient, 20 mmol.L-1) for Cefotaxime-SANAVITA-Germany, Cefotaxime-LDP-Spain & Cefotopox-Russul-India respectively. The powder was dissolved in distilled water. The solution was filtered to remove any undissolved residue that affected the response and complete the volume to 100 mL with distilled water.
Methodology
A two-line manifold reaction design was used and connected with ISNAG-fluorimeter instrument to estimate the cefotaxime sodium via its reaction with VOSO4 to form white precipitate, as shown in Figure 2. First line as a carrier stream of distilled water carried the sample segment (140 μL) of 8 mmol/L of Cefotaxime Sodium concentration at 1.8 ml/min flow rate then combined with the second line (VOSO4 Line) at 1.8 ml/min flow rate via Y-junction point that carried the reagent of VOSO4 at 7 mmol/L. So, when the reagent (precipitating agent) joined with the incoming drug causing the formation of small crystalline precipitate particulate, followed by the measurements of divergent light via irradiating the precipitated particulate by two wavelengths at 184 nm & 253.7 nm (Prominent) through homemade ISNAG - Fluorimeter instrument (Figure 2). The proposed probable reaction pattern of ion pair is shown in scheme 1 [11,12].

Figure 2: Flow diagram of used manifold throughout this part of research work

Scheme 1: Proposed reaction between CFTS with VOSO4 to form ion pair
Optimization of reaction pattern parameters
Chemicals parameters, mainly concentration of reagent and type of carrier stream for the reaction of CFTS with VOSO4 system, as well as physical parameters including sample volume, flow rate and reaction coil were studied using two-line manifold system.
Chemical variables
Effect of vanadium oxide sulphate
Variable concentrations of vanadium oxide sulphate reagent ranging 1-10 mmol/L were prepared at 140 μL sample volume, which were injected on a carrier stream (distill water). 8 mmol/L concentration of CFTS was used with 1.8 ml/min flow rate for each line. Figure 3.A shows a kind of response versus time of profile (YZi(mV)- tmin (dmm) 1(10)). The increase of VOSO4 led to an increase in the peak height reaching the value of 5 mmol/L (1552 mV). This increase might be attributed to the increase of small nuclei density which in turn start to grow to form granules as the reflecting light surface toward the detector in a more intense and shorter frequency (blue shift) and falls within solar cell limit of sensing while more than 5 mmol/L causes low sensitive responses due to the possible formation of vacancies or pockets to accommodate unwanted particulate i.e., impurities relative to the nature of precipitate causing spreading or dispersed particulate leading to decrease of diverged light toward the solar cell. This was noticed using slope-intercept method (Figure 3.B); which shows that the selected segment giving the highest value is a3 – a5 in which 5 mmol/L fell within its boundaries. Also, any chosen concentration of VOSO4 within this segment can be used as an alternative to use in newly developed methodology of optimization.

Figure 3: Variation of VOSO4 concentration on
A: Profile of CFTS – VOSO4 system
B: Height of ȲZi(mV) energy transducer response in mV with three data points as one segment their interaction and choice
Type of media effect (salt and acid)
Cefotaxime Sodium (8 mmol/L) reaction with VOSO4 (5mmol/L) was studied in different media (CH3COONH4, NaCl, Na2SO4, KBr, NaNO3, NH4Cl, KCl, HCl, CH3COOH, H2SO4, HNO3, Tartaric acid (C4H6O6) and Ascorbic acid (C6H8O6)) at 50 mmol/L concentration in addition to aqueous medium to be used as a carrier stream at 1.8 ml/min flow rate to improve the light scattering on precipitate particulate surface. It was found that an increase in S/N- response using H2SO4 compared with the use of distilled water and salts medium as a carrier stream, which might be due to the increase of particles size that will act as a reflecting surface at 0-9- and increase of the scatter of light. Therefore, H2SO4 was chosen as the best carrier stream for the next studies.
Effect of sulfuric acid concentration
Using cefotaxime sodium (8 mmol/L)-VOSO4 (5 mmol/L) system, we prepared 140 μL sample volume at 1.8 ml/min flow rate for both lines, and a series of sulfuric acid solution (10- 100 mmol/L) as carrier stream (Figure 4 and Table1). It was found during working with acids (sulfuric acid) an increase of sulfuric acid concentration more than 10 mmol/L caused a decrease in peak height of response. This could be attributed to the penetration of sulfuric acid to the precipitate depth during its crystal growth forming a soft particulate, which caused a decrease the light scattering and its deviation toward the sensor (detector in ISNAG-Fluorimeter instrument). Therefore, 10 mmol/L of sulfuric acid was chosen as optimum carrier stream for this research. When applying slope-intercept, in spite of the values of a= 1826.27 mV which represented the lowest value, it was chosen as the most appropriate that would represent the first segment for the range 10-50 mmol/L. Also, it was noticed that the excess of acid added would dissolve the precipitate completely or in step wise by increasing the acid concentration. Accordingly, higher values of intercept which represented the segment 2 and segment 3 were neglected. So, the first segment was chosen, having the lowest value for acid concentration.

Figure 4: Effect of variable concentration of H2SO4 solution on: Output of (S/N) energy transducer response in mV with chosen segment

Physical variables
Variation effect of flow rate
Fixing cefotaxime sodium (8 mmol/L)-VOSO4 (5 mmol/L)-H2SO4 (10mmol/L) system as optimum parameters and variable flow rate from (0.7 – 2.8 ml/min) for each line (carrier stream line and reagent line) was used for the study. It was noticed that at slow flow rate (0.7 ml/min), its presence in the measuring cell, which in turn caused a display at the base (ΔtB) and apex of the profile, led to a big particle, considered too heavy to deal with the movement of carrier stream through flow cell, causing a delay and elongation (Figure 5). Next, increasing flow rate reaching to 1.8 ml/min led to an increase in height of responses were obtained, that might be attributed to the increase of diverging of light directed towards the detector while more than1.8 ml/min caused a decrease in the peak height of responses, most probably due to unavailability of sufficient time for crystal to grow to higher size particulate that would work as a reflecting mirror for the incident light. Using slope -intercept method showed that the selected segment giving highest value was (a4 - a6) in which 1.8 ml/min fell within its boundaries. Also, any chosen flow rate within this segment could be used as an alternative to use in newly developed methodology of optimization

Figure 5: Effeect of flow rate on :A-Response profile ȲZi(mV)
Variation effect of sample volume
Variable volume of sample loop ranging 40 μL to 175μL by changing the length of the sample loop within the injection value while keeping all other variables are constant (i.e. CFTS 8 mmol/L)- VOSO4 (5 mmol/L)-H2SO4 (10 mmol/L) system and 1.8 ml/min flow rate. Table 2 shows the increase of sample volume to 175 μL leading to increase of peak height and giving a regular profile of the diverged incident of light while more than 175 μL led to a decrease of peak height with a wider Δt of response. So, the sensitivity of measurements was small at more than 175 μL sample volume. This result was proven through slope- intercept method in which the segment no.2 is the most suitable, that could be used to obtain sufficient acceptable result compromised with the other variables that were chosen.

Effect of delay reaction coil on S/N response profile
Using the other optimum chemical and physical parameters, variable reaction coils from 0 (without)-236 μL connected after Y- junction point directly in manifold design system were studied. It was noticed using coil at volume 78.5 μL led to an increase in sensitivity and peak height of response up to 78.5 μL, while at more than 78.5 μL it led to a decrease of diverged light with increase of base width and departure time of sample segment from injection valve reaching the measuring cell. This might be attributed to an increase of diffusion and dispersion of precipitate particulate segment causing loss of some of the reflecting surface. Therefore, delayed reaction coil at volume 78.5 μL was used in this research. This result corresponds with slope – intercept method which shows that the selected segment giving the highest value of intercept is a2-a5 (i.e., segment no.2), in which any point within it can be used to obtain acceptable results and high sensitivity (figure 6).

Figure 6: Variation effect of Coil volume on: Height of ȲZi (mV): (S/N) energy transducer respomV, three segments (Three-point data) and chosen segments
Studying the variation of energy transducer output of diverged light versus cefotaxime sodium concentration
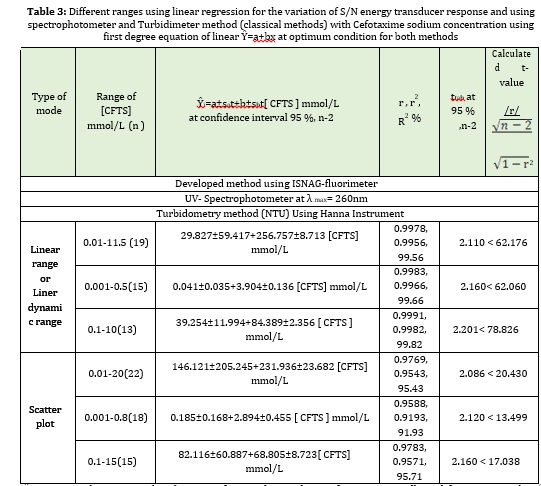
Using fixed parameters optimization achieved at previous section, we prepared a set of CFTS solutions (0.01-20 mmol/L) for CFTS-VOSO4 (5 mmol/L)-H2SO4 (10 mmol/L) system, in which the direct proportion up to 20 mmol/L between the variation of concentration of precipitate particulate formation and output of diverged light was expressed as an Zi (mV). Many variables, such as refraction, absorbance, reflection, and divergent light from inside the precipitated particles, might have increased as the beam of light diffused inside the particles. All values were measured at an angle 0-90 and represented by scatter plot at range 0.01-20 mmol/L, in which we gave a coefficient correlation of Zi (mV) versus tmin (dmm)1(10) of 0.9769 with coefficient of determination of 0.9543 and linear dynamic range 0.01-11.5 mmol/L. A short range that should be used to improve the assessment of mathematical formulation and the best Linear equation with the correlation between cefotaxime sodium concentration against diverged light as a dependent variable with r=0.9978 and % capital R-squared of 99.56% (table 3), and the linear equation of the form. ŶZi =29.827±59.417+256.757±8.713 [CFTS] mmol/L was able to explain much of obtained results by 99.56% from n= 19 as the outcome of linear dynamic. The assessment evaluation of the new developed methodology for the determination of cefotaxime sodium was compared with available references of the two methods including UV-Spectrophotometric method and turbidimetric method based on:
1- UV. Spectrophotometric method: using a quartz cell to detect absorbance for the range of correlation concentration 0.001-0.8 mmol/L at max = 260 nm [0.15 mmol/L]. With a correlation value of 0.9983 and a percent capital square of 99.66 percent, the scatter plot and best linear range ranged from 0.001-0.5 mmol/L, n=15 (no.of measurements). The variable data treatments are shown in Table 3;
2- turbidometry method; which is based on the reaction between VOSO4 as a precipitating agent with optimum concentration 6 mmol/L in aqueous medium and the drug of cefotaxime sodium for a variable range of concentration 0.1-15mmol/L. The results are tabulated in Table 3 at confidence level 95 % using first degree equation of the form of:
Ŷi(NTU) = a + b [CFTS] mmol/L

The best linear ranges extend from 0.1-10 mmol/L of n= 13 with correlation coefficient of 0.9991 and % capital R-squared = 0.9982 % (Table 3).
Limit of detection (LOD)
The Limit of detection of cefotaxime sodium is calculated by three different approaches depending on the practical research, i.e. developed method using VOSO4 (5 mmol/L) -H2SO4 (10 mmol/L) system, 175 μL sample volume and two classical methods (UV- Spectrophotometer & Turbidimetry) (Table 4).

Repeatability
The reproducibility of the determination of cefotaxime sodium was investigated using an ISNAG-fluorometer to detect the divergence of incoming light, which was generated by the reaction of cefotaxime sodium with VOSO4 in the presence of H2SO4. The relative standard deviation is expressed as a percentage (RSD percent 0.23 percent), which is proportional to the measurement's repeatability. In optimal parameters, eight consecutive injections of CFTS at fixed concentrations (2 and 5 mmol/L) were assessed. All of the information gathered is summarized in Table 5.

Determination of Cefotaxime sodium in drugs using a homemade of ISNAG-fluorometer analyzer
Cefotaxime sodium was determined in three distinct samples of medicines from three different firms using the newly developed technology (ISNAG-fluorometer). (Cefotaxime-1000mg-SANAVITA-Germany, CEFOTAXIME -500 mg-LDP-Spain and CEFOTOPOX- 500mg-RussuL-India). The continuous flow injection was analyzed by homemade ISNAG-fluorometer analyzer, in which mercury tube lamp was as a source and four solar cells at each two side (0-90) as a detector; this was compared with two classical methods including UV-Spectrophotometric via the measurements of absorbance at λmax = 260 nm and turbidometry-method via turbidity-meter- HANNA-Taiuan. Each method depends on the preparation of each drug: 20 mmol/L equivalent to 0.4774 g of active ingredient in 50 ml by transferring 1 ml to each of five volumetric flasks (25 ml), followed by addition of gradual volumes of standard solution (50 mmol/L) of Cefotaxime sodium (0, 1, 1.5, 2 and 2.5 ml) to obtain 0, 2, 3, 4 and 5 mmol/L concentration.
Table 6. A and B show the results mathematically treated [3-5], using two or three different methods, i.e. developed method, UV-Spectrophotometric method and turbidometry method, and with a realistic active component content at a 95% confidence level and determination efficiency in addition to t-test.
First test: We used individual t-test to compare the means of practicality of active ingredient (using newly developed methodology) with claimed value (i.e., μ= 500 mg or 1000 mg) as shown in Table 4.B (Column 5)
Null hypothesis: There is no significant different between the means (i (SANAVITA, 1.0324g, Germany), (LDP, 503.265 mg, Spain), (RussuL, 488.998 mg, India) (average weight)) obtained from three source of three different companies and claim value [15, 16] (μ=500mg or 1000 mg).
There is a substantial discrepancy between the means and claim value in the Alternative hypothesis.
From the results obtained, it seems that there was no significant difference between measured mean values (i) compared with official Quoted value (μ500 mg or 1000 mg) except sample no.3 possibly due to the interferences that cause a random error that exist with this sample (i.e., sample no.3).

Second test: We used paired t-test at α= 0.05 of three drugs from different manufacturing. The comparison was made between developed method using ISNAG-fluorimeter analyzer (depending on scattered light ± 90) and turbidimetry via turbidity-meter, HANA, (Taiuan) as well as UV-spectrophotometer at λmax= 260 nm. The results of comparison are summed up in Table 6. B (Column 6).
The Assumption as follows [13,14]:
Null hypothesis:
H0: μISNAG-fluorimeter = μUV-spectrophotometry = μturbidimetry
There is no significant difference between the means of turbidity method and ISNAG-fluorimeter analyzer
Alternative hypothesis:
H1:μISNAG-fluorimeterμUV-spectrophotometry μturbidimetry
As shown in Table 4.B (column 6) due to tcal less than ttab(4.303), there were no significant differences between the developed method and two classical methods (UV-spectrophotometric and turbidimetric methods) for determining cefotaxime sodium in pharmaceutical drugs at 0.05 (95% percent confidence level).

Conclusion
A new turbidimetric, simple, sensitive, accurate and fast method for determination of cefotaxime sodium in pharmaceutical drugs (tablets) was developed using a newly developed homemade ISNAG-fluorimeter -CFIA. The comparison between this works with classical turbidometry method via the t-test (as the comparison tools) showed that with no doubt the newly developed method (ISNAG-fluorimeter procedure) was as good as the classical method.
Acknowledgments
I would like to express my deepest gratitude to Prof. Dr. Issam M.A. Shakir Al-Hashimi for his appreciable advice, important comments, support and encouragement.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' contributions
All authors contributed toward data analysis, drafting and revising the paper and agreed to be responsible for all the aspects of this work.
Conflict of Interest
We have no conflicts of interest to disclose.
HOW TO CITE THIS ARTICLE
Zahraa Dhahir. Nagham Shakir Turkie. New Turbidimetric Method for Determination of Cefotaxime Sodium in Pharmaceutical Drugs Using Continuous Flow Injection Manifold Design with CFTS- Vanadium Oxide Sulfate System, Chem. Methodol., 2022, 6(2) 91-102
DOI: 10.22034/chemm.2022.2.2
URL: http://www.chemmethod.com/article_140435.html
ORCID
Zahraa Dhahir:
- British Pharmacopoeia. 7th edition. The Stationery office, Londone. 2012 [Publisher]
- Katzung B.G., Basic and Clinical Pharmacology. 2nd ed., Appleton and Lange. 1987 [Crossref], [Google Scholar], [Publisher]
- British Pharmacopoeia. London. Her Majesty’s Stationery Office. 2007 [Google Scholar]
- Turkey N.S., Al-Awadie, Ahmed F.K., Kerbala uni., 2013, 9:153 [Google Scholar], [Publisher]
- Turkie N.S., Al-Awadi, Munshid H.Q., Pharm. Sci., 2019, 11:1563 [Google Scholar], [Publisher]
- Nagham S.T., Mustafa K.K., J. Res. Pharm. Chem., 2016, 6:759 [Publisher]
- Al-Awadie N.S.T., Ibraheem M.H., , J. Res. Pharm., 2016, 6:230 [Google Scholar]
- Al-Awadie N.S.T., Khudhair A.F., Iraqi J. Sci., 2014, 55:12 [Google Scholar], [Publisher]
- PATENT, No. 3884, Shakir I.M.A., Al-Awadie N.S.T., ISNAG Fluorimeter For Fluorescence Measurement At The Visible Region Of Electromagnetic Radiation Using Mercury Tube Lamp As A Source For Irradiation For Continuous Flow Analysis Using Solar Cells. Iraq, International classification, 2014, H01J61/00
- Al-Awadie N.S.T., lbraheem M.H., J. Res. Pharm. Chem., 2016, 6:891 [Google Scholar]
- Jordan W., van Barneveld H., Gerlich O., Kleine‐Boymann M., Ullrich J., Phenol. Ullmann's encyclopedia of industrial chemistry. [Crossref], [Google Scholar], [Publisher]
- Carey F.A., Organic Chemistry. 11rd Ed., McGraw-Hill, New York.1996 [Publisher]
- Murdoch J., Barnes J.A., Statistical Tables: for Science, Engineering, Management and Business Studies. Macmillan International Higher Education. 1986 [Google Scholar], [Publisher]
- Miller J.M., Miller J.C., Stastical and chemometric for analytical chemistry. Fifth Edition. Person eduction limited 2005 [Google Scholar]
- British pharmacopoeia, Published by the stationary office London. 2003 [Crossref], [Publisher]
- The British Pharmacopoeia Commission Secretariat. . Part of the Medicines and Healthcare products Regulatory Agency (MHRA). British Pharmacopoeia, Her Majesty's Stationery Office, London, UK2009 [Google Scholar]


