Document Type : Original Article
Authors
1 Macromolecular Physical Chemistry Laboratory, University of Oran 1 Ahmed Ben Bella, Oran, Algeria
2 Department of Sciences and Technologies, Faculty of Applied Science. University Ibn-Khaldoun of Tiaret, Algeria
3 Synthesis and Catalysis Laboratory, Ibn-khaldoun University of Tiaret, Algeria
Abstract
Fluorocarbon surfactant containing perfluorinated is the key component in aqueous film-forming foam extinguishing agent (AFFF), although its environmental concern is becoming more evident. In order to reduce the amount of this fluorinated surfactant while maintaining the micellar and surface properties, mixed systems based on fluorocarbon non-ionic and hydrocarbon cationic surfactants have received more attention with the aim of decreasing the environmental hazard of perfluorinated surfactants. The micellar properties, surface activity, and synergistic behavior of mixture systems of non-ionic fluorocarbon surfactant (undecafluoro-n-pentyl-decaoxyethylene ether (C5F11EO10)) and three cationic surfactants with different alkyl chain (n-alkyltrimethylammonium bromides: 10 (DTAB), 14 (TTAB) and 16 (CTAB)) are investigated. The effect of cationic chain length on physicochemical and thermodynamic properties are evaluated. Results obtained highlight a remarkable effect of reduction of the critical micellar concentration (CMC) with an increase in length of the chain; this effect is due to high the increase of hydrophobicity with the increase of the chain length. Furthermore, mixed system (C10TAB-C5F11EO10) has optimal synergy for minimizing surface tension. Also, the case of mixed micelles solutions rich on C5F11EO10, CMC has less steric hindrance among hydrocarbons and fluorocarbons, which are more or less similar to carbon chains. Finally, the C5F11EO10 suggests a synergy of CMC reduction with intermediate activity between C16TAB-C5F11EO10 and the C10TAB- C5F11EO10. Mixed systems (Foam) containing short-chain fluorotelomers tend to promote fewer toxic alternatives.
Graphical Abstract
Keywords
- Fluorocarbon non-ionic surfactant
- Surface tension CMC
- Mixed system
- Synergistic behavior
- Foam property
- Theromdynamic models
Main Subjects
Introduction
As surfactants are present in liquids and solvents, they strongly influence their surface chemistry. Physicochemical properties of such agents such as viscosity, surface tension, and surface tension of liquids as well as chemical reactions that take place within the solvents are altered by the presence of these agents at a liquid interface [1]. Surfactants are therefore used in a wide range of chemical reactions as catalysts. In a certain reaction state (critical micelle concentration CMC), surfactants form nano-aggregates called micelles when they are sufficiently concentrated. Micelles may be used for solubilization, emulsification, drug delivery, and possibly even for the development of many food and skin care products [2, 3]. Fluorocarbon surfactant (FS) has a hydrophilic head and a fluorinated tail. It is one of the most important special surfactants due to theirs excellent properties, such as a good stability [4], high surface activity [5], ability to lower the surface tension of water, and synergetic effect [6]. Also, FS is frequently used in industrial applications where traditional surfactants are insufficient [7].
FS is a key ingredient in aqueous film-forming foam extinguishing agent (AFFF) [8]. In fact, fires caused by flammable liquids or gases pose a serious threat to life because they cannot be extinguished easily by water, which can instead cause the fire to spread [9]. AFFFs are effective at suppressing fires because they form an aqueous layer which keeps burning material from contacting oxygen and cools it [9]. Also, because of their outstanding performance as film formers, high surface activity, oleophobicity, and vapor sealants, FS have been widely used as components in many products [10]. FS based on perfluorooctanoic acid (PFOA) and perfluorooctane sulfonate (PFOS) have also been widely used in conventional processes of fluoropolymers manufacture [11]. However, these small molecules, containing fluorinated segments, can still contaminate environment due to their great stability and hence make it difficult to degrade in a long-term [12]. It is extremely hard to substitute the fluorocarbon surfactant directly [13]. In recent years, it has become necessary to reduce the amount of fluorocarbon surfactant by increasing research on surfactant mixtures, while also developing inherently safer alternatives, protecting and enhancing human health as well as the environment. Moreover, the toxicity and adverse effects of fluorinated processing aids on humans and the environment are serious concerns, and it is therefore challenging to develop alternatives that have low environmental effects, are non-accumulative, and degrade rapidly [14]. At the moment, two effective methods reduce the number of carbon atoms in the perfluorocarbon chain to C8-C10 and branching the perfluorocarbon chain [15-17]. The majority of these studies, however, have focused mostly on theory, with little attention being paid to application performance.
As a result, it was critical to select and prepare fluorocarbon surfactants with excellent performance and low cost in order to make AFFF widely used. The most effective method is to study the mixing of fluorocarbon surfactants and hydrocarbon surfactants in order to reduce the dosage of fluorocarbon surfactants in the actual formulation [18, 19]. Research has recently focused on the mixtures of fluorocarbons and other surfactants in order to develop a chemical substitute for fluorocarbon surfactants [20, 21]. Also, stronger synergistic interaction effect in mixtures has been reported. Thus, the mixed system of fluorocarbon and hydrocarbon surfactants have been extensively used in many industrial fields, such as wetting agents, firefighting and detergents [19, 22].
In this study, we have focused on the effect of chain length of hydrocarbon on the properties of synthetic binary mixture of hydrocarbon-fluorocarbon, including surface properties, aggregation behavior and the micelles shape and size. This study can provide guidance for the scientific application of mixed system of fluorocarbon and hydrocarbon surfactants by optimizing the length of the hydrocarbon surfactant chain which gives the most synergistic effect.
Materials and Methods
Synthesis of mixed surfactant with different chain alkyl
In this study a series of cationic surfactants alkyltrimethylammonium bromides (RTAB) and different alkyl chain length (10, 14 and 16 carbon atoms) were used including Decyltrimethylammonium bromide (DeTAB); tetradecyltrimethylammonium bromide (TTAB; C14H29N+(CH3)3, Br-) and hexadecyltrimethylammonium bromide (CTAB; C16H31N+(CH3)3, Br-), which were purchased from Aldrich with 99% of purity. The FSO100 (C5F11–(OCH2CH2)10–OH, (abbreviated C5F11EO10 in what follows) was provided from Zonyl company. Solvent used was deionized water with a surface tension of 69.9 mN/m and a specific conductivity of 3 μS/cm at 25 °C. All used surfactants were soluble in aqueous medium at temperatures below 25 °C. Solutions (surfactant/ water) were prepared at room temperature, by mixing stock solutions of CnTAB (10 mM) and FSO100 (C5F11EO10) (5 mM) in varying proportions. In brief, a volume of 50 ml of mixed system was prepared by mixing two volumes of concentrated solutions of each surfactant (cationic and non-ionic) according to the desired fraction of FSO-100 at each step (0.2, 0.4, 0.6 and 0.8).
For determination of CMC, the measurements were performed on concentrated samples (stock solutions) then diluted successively. Temperature was stabilized at 25 °C during all manipulations using a thermostatic bath.
Method to Surface Tension Measurement
The measurements of surface tension (γ) were carried out using a tensiometer based on Du Noüy method and operates over a range of values ranging from 0 to 90 dyn / cm with accuracy of 0.05 dyn/ cm. The platinum ring is immersed in the solution with an ascending force applied to it. The force required to tearing of the ring is equal to the surface tension that can be read directly on the calibrated device [19, 23].
Theoretical background
Clint Model
For free monomers of an ideal mixtures of two surfactants and assuming a coefficient of unit activity, CMC of mixed surfactants can be predicted using Clint’s model, given by following relation (1):

CMC1, CMC2 are the CMC values of both pure components and X1 is the molar fraction of surfactant 1 in the mixture.
In addition, according to the ideal state model, the mole fraction of surfactant 1 in the ideal mixed micelle ( ) was estimated using the following formula (2):

Rubingh Model
This descriptive model enables a good understanding of micellar properties and superficial based on parameters as the interaction parameter within the micelles ( m)through a simplified approach of the mixing non-ideality [24]. As for the activity coefficient, the CMC of the mixture is given by an equation similar to Clint's but incorporating the activity coefficients of surfactants 1 and 2 ( , ) in the mixed micelle:

the interaction parameter is given by:

the mole fractions of the two surfactants in the mixed micelle are calculated by solving the following transcendental equation:

Maeda model
Maeda's model proposed in 1995 [25] has explicitly introduced chain-chain interactions in addition to head-to-head interactions in a mixed ionic/ non-ionic surfactant system. This model is based on the phase separation model; micellization free energy is given by:

signifies the head-head electrostatic interaction, it is equivalent to in the Rubingh theory. It is related to by:
![]()
represents the chain-chain interaction and is associated with the change in the standard free enthalpy, accompanying the replacement of a non-ionic molecule by an ionic molecule.
Results and discussion
Pure surfactants
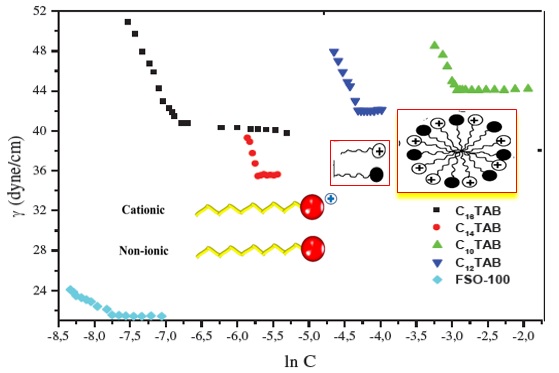
Figure 1 shows that the nonionic surfactant FSO-100 has less CMC value of γ: 21 dyne/cm. Indeed, the fluorine atoms increase the hydrophobic effect and decrease the CMC, compared with their value for cationic surfactant. Additionally, no charge on the hydrophilic head, reduce electrostatic repulsion and promoted the formation of micellar aggregates at low CMC values. Results based on CMC values (Figure 1) for alkyl ammonium bromides are better and in accordance to other studies.
Mixed systems
Critical micelle concentration CMC
Measurement of surface tension of prepared mixtures of CnTAB/FSO-100 under different molar fraction of 20%, 40%, 60% and 80% of total concentration are represented in Figure 2. The CMC values obtained for all mixtures under different fractions are regrouped in Table 1.

Figure 1: Surface tension as a function of the logarithm of the concentration of different surfactan
Table 1: The CMC values for systems: FSO-100 / HTAB, FSO-100 / TTAB, FSO-100 / DTAB, FSO-100 / DeTAB
|
X[fso-100] |
CMC(FSO-100/HTAB) mM |
CMC(FSO-100/TTAB) mM |
CMC(FSO100/DeTAB) mM |
|
X=0 |
1.070 |
3.356 |
53.510 |
|
X=0.20 |
0.128 |
0.390 |
0.340 |
|
X=0.4 |
0.137 |
0.250 |
0.362 |
|
X=0.6 |
0.148 |
0.240 |
0.403 |
|
X=0.8 |
0.143 |
0.180 |
0.360 |

Figure 2: Surface tension as a function of the logarithm of the total surfactant concentration [mM] for pure FSO100 and the 3 systems DeTAB / FSO-100, CTAB / FSO-100, TTAB / FSO-100 with a), cosurfactant molar fractions of 0.20. b) of 0.40, c) of 0.60, d) of 0.80
As shown in Figure 2, the CMC of the different synthesis mixtures is lower than that of pure surfactant, indicating the presence of synergy on micelle formation (type I). Except for the case of a 20 percent ration, we found a decrease in CMC for FSO-100/CnTAB depending on the length of the hydrocarbon chain of the added cationic surfactant. This decrease in CMC can be explained by the effect of the hydrophone on molecular aggregation formation. However, for a 20 percent FSO-100 ratio, the CMC of mixture FSO-100/C10TAB is lower than that of mixture FSO-100/C10TAB. It has also been observed that the decrease in CMC values of cationic surfactant (CnTAB) in the presence of non-ionic surfactant may be due to incorporation of the lowest proportion, in this case 20%. Regardless of the length of the CnTAB chain, few enrichments are observed for the CMC in the rest of the ratios mixture.
Interfacial pressure
Figure 3 shows the variation of superficial pressure πCMC as a function of fluorocarbon surfactant mole fraction XFSO-100. πCMC of adsorbed film is defined as the difference between the surface tension of pure water*0 and the surface tension of the solution at CMC:
γmin (π CMC = γ0–γmin).
According to the results obtained, cationic FSO-100 is more efficient than pure surfactant CnTAB due to its combination with nonionic FSO-100. The effect is most evident when going from 0 to 20% then remaining relatively constant, especially for C10TAB/FSO-100 where the surface pressure is optimum at XFSO=0.2 as this indicates a synergistic effect.
As shown Figure 3, the data obtained for interfacial pressure indicate a possibility to plot the superficial pressures πCMC versus XFSO-100. Also, the πCMC of adsorbed film (formed on the surface) is defined as the difference between the surface tension of the pure water γ0 and of the solution CMC:
γmin (π CMC = γ0–γmin)

Figure 3: Influence of interfacial pressure of the adsorbed film for the three mixed systems as a function of the FSO-100 proportion
Clint model
The theoretical results obtained of CMC for ideal mixture according to Clint Model, were compared with experimental results of CMC from tensiometer measurements, of three mixtures containing the FSO-100 with 20 to 80% proportion of composition. As shown in Figure 4, systematic negative deviation from origin is represented by theoretical curve of variation of CMC of XFSO-100 for all mixtures with all proportion. This result may be due to a predominant interaction of attractive type in micellar systems between FSO-100 molecules and each catioic surfactants for all proportions and regardless of length of CnTAB chain (type I of synergy) [26].
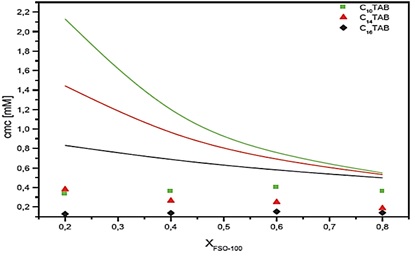
Figure 4: Experimental and theoretical critical micellar concentrations (curves) (Clint model) versus XFSO-100 for the three FSO-100 / C16TAB systems, FSO-100 / C14TAB and FSO-100 / C10TAB
We observed that the deviation of three systems is important for the lowest value of XFSO-100, and for their highly values close to ideality. Figure 5 shows the variation of micellar composition of XCnTAB as a function of total CnTAB content using Clint model. Therefore, the results indicate that micelle mixed system of C16TAB/FSO-100 is richer in cationic surfactant, but C10TAB/FSO-100 mix is less rich. These results are valid, hypothesizing an ideal mixing behaviour. This is not the case for the systems studied here as we have just shown. These data will serve as a baseline for analysing the results on the next section.

Figure 5: Hypothetical micellar composition in FSO-100 according to the Clint model (ideal mixture)
Rubingh Model
Table 2 groups the different values of , and the enthalpy free of excess micellization for all mixed systems as a function of XFSO-100.
We can see from Figure 6 the variation of ßm according to XcnTAB in all proportions. The negative values of ßm for all mixed systems CnTAB / FSO-100, may be explained by the presence of interaction with attractive type and synergistic effect for all mixture. These experimental results are consistent with those of Rosen and Col [27], which indicate a synergism type (1), since the following inequalities are verified.
Table 2: Thermodynamic parameters obtained from the Rubingh model for different values of XFSO-100 for FSO-100 / HTAB systems, FSO-100 / TTAB, FSO-100 / DeTAB
|
XFSO-100 |
0.8 |
0.6 |
0.4 |
0.2 |
|
|
0.37 |
0.30 |
0.22 |
0.18 |
|
|
0.46 |
0.40 |
0.34 |
0.31 |
|
|
0.53 |
0.47 |
0.42 |
0.37 |
|
|
-10.7 |
-8.7 |
-7.2 |
-7.2 |
|
|
-5.5 |
--5.9 |
-5.6 |
-7.2 |
|
|
-7.6 |
-6.5 |
-5.9 |
-6.6 |
|
Δ (kj/mol)DeTAB |
-6.1 |
-3.7 |
-3.7 |
-2.6 |
|
Δ (kj/mol) TTAB |
-3.4 |
-3.5 |
-3.1 |
-3.8 |
|
Δ (kj/mol) HTAB |
-4.7 |
-4.0 |
-3.5 |
-3.8 |

Figure 6: Variation of interaction parameters according to the model of the regular solution. For the three systems (a) FSO-100 / TTAB (b) FSO-100 / DTAB (c) FSO-100 / DeTAB
In comparison to the C14TAB and C16TAB systems, interactions are increasingly strong for HTAB, except for the 20% case in CnTAB. These interactions are expected to be caused by the significant hydrophobic effect present in synergistic interactions in a non-ionic-cationic mixed system. Similar results reported by Basu Ray et al. [28], where average interaction for MEGA-10/C16TAB, MEGA-10/C14TAB, and MEGA-10/C12TAB mixed systems were -1.52, -0.88, and - 0.65, respectively; hydrophobic effect is at the origin of this behavior. The deviation from ideality, on the other hand, increases as the length of the hydrophobic chain increases.
In contrast to previous behavior, the C10TAB / FSO-100 system appears with a particular behavior. In fact, strongest interaction was observed for most negative value of ßm (-10.7 for 20% in FSO-100). These values are comparable to those reported for similar systems [25, 29], where similarly the presence of strong attractive interactions entering molecules have been observed. Both surfactants linked with a powerful interactions effect. In particular, with a 20% FSO-100 Type II synergy, the surface tension at the CMC is lower than pure surfactants. Figure 7 shows the variation in as a function of XCnTAB.
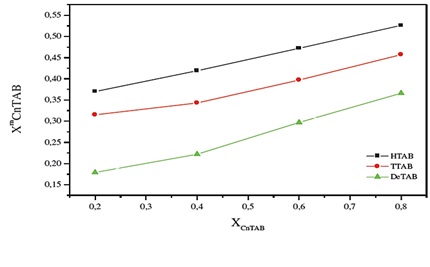
Figure 7: Variation of according to the model of the regular solution as function as XCnTABFor the three systems (a) FSO-100 / TTAB (b) FSO-100 / DTAB (c) FSO-100 / DeTAB
As shown in Figure 7, the similar behavior can be observed for three mixed systems exanimate, and a progressive enrichment of micelle CnTAB with increasing of proportion in CnTAB. we noted for a given percentage, mixed micelle is richer of cationic tension active with increasing of chain length. Moreover, for all mixed systems except in 80% in C16TAB, where it is rich in FSO-100 (XCnTAB), this behavior is due to micellization of non-ionic surfactant as easier (at lower CMC) than that for CnTAB; similar behavior has been observed by Basu Ray et al. [30].
The values of excess free enthalpy of mixed micellization have negative values as reported in Figure 8. These results indicate that mixed micellization favorite compared with each of the two pure components, for all systems and under all proportions.
For the range of 20 to 40% in FSO-100, is more negative when increasing the chain length of cationic surfactant (cases of C14TAB and C16TAB). Except for the case of C10TAB, a negative value of . Result showed a high synergy for all systems particularly those rich in FSO-100, where spontaneity of formation on mixed micelles increases with the length of hydrocarbonic chain, and the gap height is enriched with FSO-100.

Figure 8: Variation of excess free enthalpy of micellisation of mixtures
The obtained results based on Rubingh Model indicate that aggregates are strongly rich in C10TAB, due to strong attractions inter-molecular. The mixed C14TAB/FSO-100 exhibits similar behavior; however, the mixed C16TAB/FSO-100 with 20%FSO-100 is an ideal mixed surfactant, due to their composition richer on cationic surfactant. Thus, for this fraction, the mixed micelles are enriched in FSO-100. For the other fractions, the behavior is similar to that of the other mixtures.
Figure 9 presents the deviation percent of from ideality. we observed an important deviation for the mixture C10TAB/FSO-100 at 20% and less important one for mixture C16TAB/FSO. For the system with proportion more than 40% in FSO-100, the different deviations meet more or less upward (C14 and C16) or downward (C10).
This behaviour could be related to the CMC values of the pure surfactant, which is very different (case of C10TAB compared with FSO-100), that will cause a very important change in the properties of the mixture. On the other hand, if the CMC of the two pure surfactants have similar values, the non-ideality will be accentuated with the increase of the fraction of the most effective surfactant (FSO-100) [31].

Figure 9: Deviation of micellar composition from ideality as a function of XFSO-100 according to the Rubingh model
Maeda model
The different interaction parameters calculated according to equation (6) for a proportion of 20% FSO-100 are regrouped in Table 3.
Table 3 clearly shows that the value of β1 is associated with the variation in free enthalpy when an ionic surfactant is replaced by a non-ionic surfactant in the micelle. The most negative value was observed in the case of FSO-100/DeTAB system. This result is in good agreement with previous observations. In fact, this behavior is related to the greater proximity of hydrocarbon and fluorocarbon chain lengths resulting in increased of insertion of DeTAB chains (14 Å) into FSO-100 (8Å) non-ionic surfactant chains in the mixed micelle.
Thus, high disparity between chain lengths induces weaker attractive interactions, also indicating that steric chain factor plays a major role in mixed micellar formation.
Table 3: The Maeda parameter values for the three FSO-100 / TTAB, FSO-100 / DTAB and FSO-100 / DeTAB systems
|
|
DeTAB/FSO-100 |
TTAB/FSO-100 |
HTAB/FSO-100 |
|
β0 β1 β2 |
-7.73 -13.23 8.43 |
-7.73 -8.08 6.05 |
-7.73 -7.54 6.65 |
.

Figure 10: Variation of the free melting enthalpy (according to the Maeda model) according to the length of the chain for the three FSO-100 / TTAB, FSO-100 / DTAB and FSO-100 / DeTAB system
Figure 10 illustrates the variation of ΔGmic based on the length of the cationic surfactant chain at different proportions of CnTAB, for each of three mixtures and at different values of XFSO-100. Negative values of ΔGmic clearly indicate that spontaneity of mixed micellization is most important for the shortest cationic surfactants of FSO-100 mixtures (X FSO-100<50%). As we have already mentioned, the opposite results obtained for mixtures richer in FSO-100 with the longest cationic compound show greater spontaneity for the formation of mixed micelles.
Predictions of the micellar form
As is known for surfactants and mixed surfactants, determining the packing of surfactants is more difficult than determining the packing of hard substances due to the soft nature of their boundaries. The basic packing parameter P is widely used to justify the geometrical shape of these structures, which is given as : P = ; where : l is the length of the hydrophobic chain of the surfactant, a is the area per polar head and v is the volume of the hydrophobic chain [32]. In the case of a mixed system, the effective packing parameters obtained according to the following equation (10), which has been reported by several recent works [33-35]:
where l is the length of the hydrophobic chain, a denotes the surface of the head and v signifies the volume of the hydrophobic chain
Tanford formulas are also used in this work to calculate the lengths and volumes of the chains of all surfactants studied [36]. Polar head sections were deduced directly from tensiometry data from the Gibbs isotherm represented by equation [37].
Based on obtained results data of packing parameter summarized in Table 4, the micellar aggregates of TTAB/FSO-100 and HTAB/FSO-100 mixed systems are spherical shape with diameters of 40 nm, just as the type of geometry observed for pure cationic surfactants. On the other hand, the stacking parameter value of the DeTAB/FSO-100 is greater than 0.3, which indicates their preferred cylindrical shape. This value is indeed intermediary between that of DeTAB (spherical micelles) and that of FSO-100 (vesicular aggregations).
Table 4: Values of the effective packing parameter of mixed systems for different proportions in FSO-100
|
XFSO-100 |
DeTAB/FSO-100 |
TTAB/FSO-100 |
HTAB/FSO-100 |
|
20% |
0.36 |
0.16 |
0.15 |
|
40% |
0.36 |
0.21 |
0.16 |
|
60% |
0.36 |
0.21 |
0.16 |
|
80% |
0.36 |
0.22 |
0.16 |
Conclusion
To reduce the dose of fluorinated surfactant, we have improved the properties of surfactant mixtures to be considered as safer alternatives for protecting human health and the environment. A series of experiments on the physico-chemical characterizations of pure hydrocarbon cationic surfactants with different chain lengths, such as (DeTAB), (TTAB), and (HTAB), mixed with non-surfactant fluorinated ionic undecafluoro n-pentyl decaoxyethylene (FSO-100) were carried out in this work. The following are the major findings:
- A systematic decrease of CMC for the three mixed systems studied compared with pure surfactants, reflecting an increasing synergistic effect with increasing chain length.
- The mixed micellar system favours excessive incorporation of the CnTAB molecules (C10TAB and C14TAB cases).
- According to Rubingh Model, the interactions between hydrophobic chains are attractive as the chain length decreases. This behaviour shows the existence of a powerful synergistic effect linked to a less steric hindrance between the hydrocarbon chains and fluorocarbon of the two surfactants, promoting greater spontaneity of micellar formation. (case of FSO-100/DeTAB system)
- Based on the Clint and Rubingh models, the estimated micellar compositions of FSO-100 at various proportions in FSO-100 were assessed. The largest deviation is for C10TAB/FSO-100 mixture at 20%, but for C16TAB/FSO-100 mixture is less significant.
- Synergistic effect identified for C10TAB/FSO-100 and evidence of strong chain interactions in this particular case.
Finally, the predictions of mixed micellar forms indicate a cylindrical shape in the case of the FSO-100 / C10TAB system and a spherical micellar form in the case of C14TAB / FSO-100 and C16TAB / FSO-100.
Acknowledgments
My thanks and gratitude to everyone who continuous support and review of all work. We are also immensely grateful to Prof. Farida Bouanani.
Funding
This research did not receive any specific grant from fundig agencies in the public, commercial, or not-for-profit sectors.
Authors' contributions
All authors contributed toward data analysis, drafting and revising the paper and agreed to responsible for all the aspects of this work.
Conflict of Interest
We have no conflicts of interest to disclose.
ORCID
Mohamed Naous
https://www.orcid.org/0000-0003-0025-4994
Ahmed Halfadji
https://www.orcid.org/0000-0002-4059-4910
Abdelkader Chougui
https://www.orcid.org/0000-0001-6450-9277
Boumediene Bounaceur
https://www.orcid.org/0000-0002-9200-0790
HOW TO CITE THIS ARTICLE
Mohamed Naous, Ahmed Halfadji, Abdelkader Chougui, Boumediene Bounaceur. Micellar Properties of a Foam Extinguishing Agent Based on Fluorocarbon Non-Ionic and Hydrocarbon Cationic Surfactants in a Mixed System. Chem. Methodol., 2022, 6(7) 534-547
|
HOW TO CITE THIS ARTICLE
Mohamed Naous, Ahmed Halfadji, Abdelkader Chougui, Boumediene Bounaceur. Micellar Properties of a Foam Extinguishing Agent Based on Fluorocarbon Non-Ionic and Hydrocarbon Cationic Surfactants in a Mixed System. Chem. Methodol., 2022, 6(7) 534-547
|