Document Type : Original Article
Authors
Department of Chemistry, College of Science, University of Baghdad, Iraq
Abstract
In the current work, N-substituted cis-1,3,4,6-tetra hydrophthalimide polymerized derivatives were prepared as bio-based materials. This research was carried out through the following three steps; Firstly, preparing N-alkyl cis-1,3,4,6-tetra hydrophthalamic acid by reacting of cis-1,3,4,6-tetra hydrophthalimide salt with (benzyl chloride, PVC, adipoyl chloride, co-poly vinyl acetate chloride and poly acyloyl chloride) respectively producing, N-alkyl tetra hydro phthalimide. Furthermore, the next step includes preparing N-creatinyl tetrahydro phthal imide, and followed by the polymerization step of the prepared unsaturated compounds throughutilizing (BPO) as initiator alongside the heating. Several techniques were used to characterize the prepared compounds such as recording the melting points and softening point as well as infrared spectroscopy (FTIR). However, some of the prepared compounds were characterized by using proton nuclear magnetic resonance 1HNMRand studied thermally via differential scanning calorimetric technique DSC.
Graphical Abstract
Keywords
Main Subjects
Introduction
Bio-based resins which usually called bio-based polymers are obtained from renewable resources (plants, microorganisms, bacteria, algae, etc.). The synthesis of these materials are carried out either directly or by synthesizing the monomers which undergo polymerization. There are different types of bio-based polymers that are available in the market such as poly-L-lactide, polylactic acid, polyhydroxybuturate, polyhydroxyalkalonates (PHAs), polyamide and polypropylene (PP) which can be obtained from bio-based ethylene which can be acquiredthrough converting thermoplastic materials such as terephthalate, ethanol, polyethylene . Other bio-based resins such as (bio based epoxy epichlorohydrine and polyurethanes with bio-based dyols) which can be obtained partially from glycerol. While the most known applications for bio-based polymers are in the field of packaging, still their uses in other demanding fields are of a great challenge. With taking the following evidences into consideration [1], our plant and the living organisms can take the advantage of using bio-based polymers as they can be used in decreasing the carbon footprint due to the fact that fungi and bacteria (microorganisms) have the ability to degrade these polymers to their constituent monomers and producing CH4, CO2, and water, therefore, no toxic materials will be generated. Moreover, the bio-based plastics have a wide industrial and daily life applications, in response to increase of the world’s population and use of technology [2].
Cyclic imides are one of the pharmacophores which considered to be an important building blocks for synthesizing agrochemicals, drugs, natural products, advanced materials and polymers. Lamprolobine, julocrotine, migrastatin, salfredin C-1, cladoniamide A, and palasimide are some of natural products that possess the imide moiety [3-6]. Imides can be used extensively to synthesize different bio-active natural products [7-13]. For example, the structural feature of phthalimide includes –CO-N(R)-CO- as well as imide ring which unable the compound to be used in terms of biological and pharmaceutical activity [14]. Huge attention has been directed toward phathalimide and its derivatives due to the proven biological effects that are even better than the known pharmacological molecules, some of the biological effects are; anxiolytic [18], antitumor [16-17], analgesic [15], antifungal, and antibacterial properties. Therefore, the biological activities of these molecules have been subjected to a lot of biomedical research [19-21].
When it comes to synthesizing phthalimide derivatives, Gaberiel synthesis method is considered the main method for that purpose. This method includes N-alkylating the potassium or sodium salt of phthalimide with primary alkyl halide in order to produce the corresponding N-alkyl phthalimide [22-23]. Most of the secondary alkyl halides cannot be involved in the reaction. In addition, most of the electrically conductive and high strength polymers are containing imide subunits such as polyimide. Kapton is one of the example of these polymers where two imide groups are contained in the repeated unit which derived from aromatic tetra carboxylic. Acids [24-27]. Polyglutarimide represents another example of polyimides which typically derived from ammonia and primary amine and poly methyl methacrylate (PMMA). The rigidity of imide functional groups is influencing the toughness of these materials [28-30]. Aminolysis and cyclization processes of PMMA is carried out at high pressure and temperature.
Materials and methods
Chemistry
Electro thermal capillary apparatus was used to determine the melting points of the prepared compounds and the values are uncorrected. Perkin Elmer IR spectrophotometer was used to record the infrared (FTIR) spectra with (4000-400 cm-1) range of wavenumbers utilizing potassium bromidepellets. Moreover, Bruker AC-400F, 400 MHz was used to perform proton (1H) nuclear magnetic resonance spectroscopic analysis. Differential Scanning Calorimetry (DSC) was used to study the thermal behavior of the prepared compounds.
1-Preparation of N-alkyl tetrahydrophthalimide (1-5)
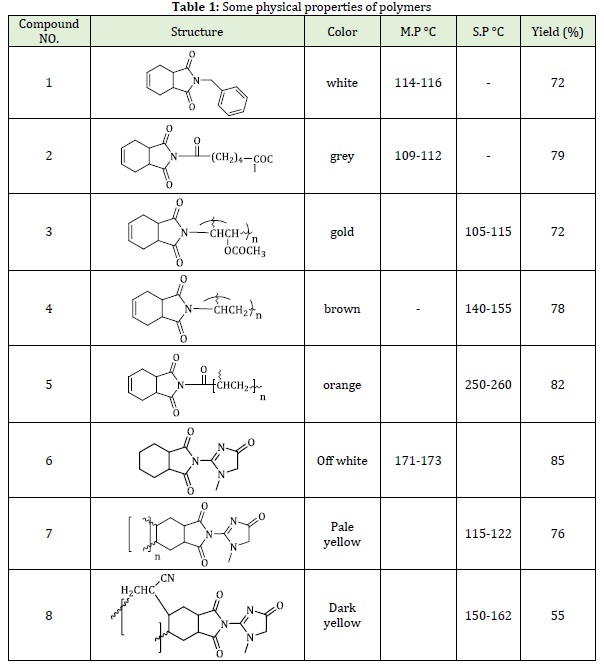
To a solution of tetrahydro phthalimide salt (0.01 mol, 1.48 g) and 25 mL of acetone was added 0.01 mol of benzylchloride (PVC, or adipoyl chloride, or co-poly venyl acetate chloride or polyacyloyl chloride) respectively [28]. Moreover, for two hours, the stirring was continued at room temperature and then filtrate the resulted precipitate followed by drying and recrystallizing from ethanol. Some physical properties of compound (1-5) are listed in Table 1.
2-Synthesis of N-creatinyltetrahydrophthal imide [31]
25 mL of a dry acetone and 0.005 mole (0.5 g) of acetic anhydride were placed in a 150 mL two-necked flask which connected to droop funnel and reflux condenser and equipped with magnetic stirrer. By stirring, all the acetic anhydride was dissolved and then 0.005 mole (0.5 g) creatinine solution in 25 mL of acetone was allowed to run through the dropping funnel drop wise for 30 min under cooling range (0-5 °C). The stirring was continued for 2 h under room temperature. Suction filtration was used to separate and filtrate the off-white precipitate. After that, the precipitate was washed with solvent and then dried and recrystallized from ethanol, the 85% yield was obtained pale brown color with m.p. 171-173.
3-polymerization of N-creatinyltetrahydrophthal imide [31]
5 ml of DMF was used to dissolve 0.3 g of the prepared monomer, and then adding 0.05 % weight of dibenzoyl peroxide. Under nitrogen atmosphere, the water bath was covering the polymerization tube in order to be heated to 900C for1 hr. The polymer was obtained as a pale yellow with 76% yield and the S.P 115-124 °C as listed in Table 1.
4-Polymerization of N-creatinyltetrahydrophthal imide acrylonitrile (co-polymerization) [8]
From prepared monomer (0.3 g) and acrylonitrile, where the DMF was used as a solvent as both compounds dissolved in 15 ml of the solvent, and then adding 0.05% weight of dibenzoyl peroxide. Under nitrogen atmosphere, the water bath was covering the polymerization tube in order to be heated to 900C for 1.5 hr. The co-polymer was obtained as a dark yellow with 55% yield and the S.P 150-162 °C. The some physical properties of compound (6-8) are listed in Table 1.
Swelling studies
The study of polymer swelling was conducted as follow: the swelling and diffusion parameters was determined by swelling the homo and copolymer in a solution with pH=7 at 37 °C. The swollen matter was removed regularly from the water bath and then superficially dried using filter papers.
Differential scanning calorimetry (DSC)
Perkin-Elmer Pyris I analyser was used to record DSC scans at 10 °C/min rate of heating, whereas, the Scanning Calorimetry of (DSC)Analysis was differential indium which used as calibration standard.
In fact, DSC is considered as a technique for determining the absorbed or released heat quantitively when a physical or chemical change is occurred to the substance.
The biological activity [32,33]
Biological activity of the synthesized compounds was tested, as some of the compounds have shown antimicrobial activities when exposed to gram negative (E. coli) as well as gram positive (Staphylococcus Aurous). The disc diffusion method was used to study the antimicrobial activities for the synthesized compounds as the bacterial inoculums were spread on nutrient agar. After drying the inoculums, wells of 8 mm diameter were made inside the plate of agar alongside the sterile cork borer. DMSO (10-3 M) concentration was used as a solvent for the synthesized compounds and the standard compound for antibacterial activity was cephalexin (10-3 M). The Petri was subjected to incubation at 37 °C for 24 h and the inhibition zone was measured in millimeters.
Results and Discussions
Gabriel synthesis method was used to syntheses all the prepared compounds, as the reaction steps have been show in the Scheme 1.
Whereas compounds 6, 7 and 8 were prepared as shown in the Scheme 2.
The starting material of the substituted synthetic compounds of N-imide was tetrahydrophtjalimide and salt’s which reacted with halogen atom of compounds at different °C, where compounds 6 and 7 was polymerized with BPO in DMF. All compounds characterized by physical and chemical properties with FTIR and H-NMR, C-NMR some of them.
FT-IR showed disappearance of uNH group band at 3203 cm-1, whereas a significant absorptions at the regions 1770-1780 cm-1 of uC=O imide group where showed, and bands at 1600-1647 cm-1 which belongs to uC=C Imide, 1883 cm-1 of ester compound 3, 1708-1670 cm-1 of uC=N creatinyl ring, 1327-1370 cm-1 of uC-N and 1152-1263 cm-1 contributed to uC-O group. However, some additional absorptions of compounds’ functional groups have been listed in the Table 2. 1H-NMR spectra of compound 3, 5 and 8 showed signal at d(0.83-0.92) ppm for (s, 5H, CH Polymer), d (3.1) ppm for (s, 3H, CH3 Ester), and d (5.56) ppm (s, 2H, CH=) for compound (3, 5), while compound 6, 7 and 8 show d(3, 06-3.2) ppm for (s, 3H, NCH3), d (4.37-4.42) ppm for (s, 2H, CH2 ,creatinin ring). On the other hand compound 8 show disappearance of signal at d(5.8) ppm duo to C=CH with appeared of signal at d(4.08) ppm duo to CH2 creatinine ring .Additional of essential signals for compounds shown in Figures 4,5 and 6.
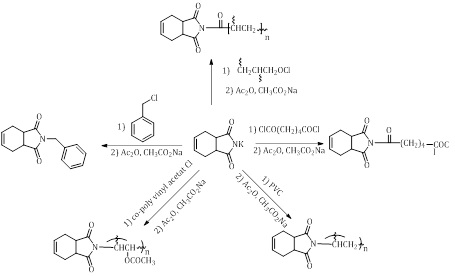
Scheme 1: Preparation of compounds 1-5

Scheme 2: Preparation of compounds 6-8
Table 2: FTIR spectral data of the prepared compounds
|
Compound No. |
C-H Olefin or Ar |
C-H Aliphatic |
C=O Imide |
C=C olefin |
C-N |
C-O |
Others |
|
1 |
3045 |
2953, 2887 |
1750 |
1618 |
1303 |
1240 |
C-C (Ar) 1445, 1567 |
|
2 |
3041 |
2910, 2888 |
1774 |
1620 |
1325 |
1267 |
COCl 1841 |
|
3 |
3015 |
2920, 2856 |
1778 |
1641 |
1329 |
1217 |
CO Ester 1838 |
|
4 |
3012 |
2902, 2889 |
1783 |
1608 |
1323 |
1265 |
- |
|
5 |
3066 |
2979, 2887 |
1769 |
1606 |
1348 |
1230 |
- |
|
6 |
3026 |
2925, 2869 |
1771 |
1600 |
1345 |
1239 |
CO cry. Ring 1708 |
|
7 |
3016 |
2969, 2877 |
1780 |
1647 |
1305 |
1244 |
CO cry. Ring 1670 |
|
8 |
3009 |
2952, 2820 |
1763 |
1632 |
1307 |
1229 |
CN 2231 |
Study of Swelling
The study of dynamic swelling for the polymer includes the following: The polymer was immersed in THF solution at room temperature and with pH=7 in order to determine the swelling and diffusion parameters. The filter paper was used to remove the swollen gels which then weighted and located in the previous bath. Moreover, dynamic swelling study was performed in order to investigate the behavior of time-dependent swelling of the polymer which included in a solution with pH=7. The following equation used to calculate the swelling percentage S%:
Where M0 represents the dry polymer mass at time=0 which equal to 0.11 gm, whereas M1 represents the swollen mass of polymer 3. at time=t which equal to 0.23 gm within 24 h it is worth noting that swelling with several days give parameter as shown in Figure 1.
Differential Scanning Calorimetric (DSC)
Polymer 7 showed 72% mass loss in 360 °C (Figure 2) while polymer 8 showed losing mass in two step one of them at 322 °C in 49% and the other at 594 °C in 47% (Figure 3). As shown in Figures 2 and 3 the TG analysis of this two polymer show thermal stability of polymer 7 seem more than thermal stability of polymer 8 that may be duo to homogenous surface effect

Figure 1: swelling with several days of polymer 3

Figure 2: DSC (TG)-analysis of homo creatinyl polymer 7

Figure 3: DSC (TG)-analysis of co-acetonitryl creatinyl polymer 8
The Biological activity
Unfortunately, there is no interesting result was found as expected, all compounds show negative or little more than cephalexin cooperative. This result might contribute to the materials used in examination test which might be expire.
Table 3: Biological activity of some of the prepared compounds
|
Compound No. |
Staphylococcus aurous |
Escherichia coli |
|
Standard cephalexin |
48 |
40 |
|
3 |
3 |
2 |
|
4 |
2 |
5 |
|
5 |
3 |
1 |
|
6 |
2 |
3 |
|
7 |
4 |
2 |
|
8 |
2 |
8 |
Note: Inhibition zone in mm
Conclusion
The prepared compounds showed thermal stability and good response mass of swollen in one case while giving negative response in the biological activity test.
Acknowledgments
Funding this research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' contributions
All authors contributed toward data analysis, drafting and revising the paper and agreed to responsible for all the aspects of this work.
Conflict of Interest
We have no conflicts of interest to disclose.
ORCID:
Muna I. Khalaf
https://www.orcid.org/0000-0002-2690-1566
HOW TO CITE THIS ARTICLE
Muna I. Khalaf, Asmaa Mohammed Noori Khaleel. Bio- Based Polymers Derived and Modified on Cis -1, 3,4, 6-Tetrahydrophthalimide and Its Applications. Chem. Methodol., 2022, 6(9) 710-719


