Document Type : Original Article
Authors
1 Chemistry Department, Payame Noor University, 19395-4697, Tehran, Iran
2 Department of Biology, Payame Noor University, Tehran, Iran
Abstract
In this study, the ligand 2-amino-3-(1-hydrogen-indol-3-yl) propanoic acid (Trp) was used to react with chromium metal ion to prepare complex [Cr (Trp)2(OH)(H2O)].This compound was synthesized for the first time and identified by elemental analysis, FT-IR, UV-Vis, conductivity, and melting point techniques. In the FT-IR spectrum of the complex [Cr (Trp)2(OH)(H2O)], the vibrational bands of the acetate and NH2 groups have shifted to lower energies than the free ligand. In addition, the presence of the Cr-N vibration band at 424 cm-1 is due to the coordination of the ligand to chromium metal. The hexa-coordinated complex of Cr with the octahedral geometric structure including [Cr (Trp)2(OH)(H2O)] is proposed.
Graphical Abstract
Keywords
Main Subjects
Introduction
Through the synthesis of organic ligands containing various donating groups, coordination chemistry is constantly expanding, and it grows in importance particularly when these ligands possess biological significance [1]. “Amino acids” are a distinct class of organic compounds and serve as components of chiral compounds (except glycine) so that they play a crucial role in biochemical processes within living organisms [2,3]. As a fundamental group of biomolecules, amino acids include amino groups (-NH2), and carboxyl (-COOH) accompanied by a specific side chain group (R: alkyl/aryl) [4]. Amino acids are functional components of protein building blocks, neurotransmitters and biosynthesis that play a crucial role in facilitating the synthesis of organic-inorganic hybrid materials [5]. N and O atoms in amino acids are the ultimate reason that complex units with different metal ions form since they are capable of functioning as oxygen and Nitrogen-donating ligands and have a crucial role in the formation of chelating rings of metal complexes [4,6-7]. “Tryptophan”, one of the twenty primary amino acids of living cells, is effective in the production of neurotransmitters and coenzymes such as “Niacin” [7] (Figure 1). Furthermore, the function of tryptophan and its endogenous metabolites, serotonin, and melatonin influence numerous biological processes [8].
Amino acid-metal interaction at the atomic level is necessary due to their application in the pharmaceutical industry and various fields of biochemistry. Recently, some metal amino acid nanomaterials which have been produced through metal-amino acid coordination, are used in drug delivery, imaging, cancer treatment, and antibacterial applications. They have shown unique advantages such as good biocompatibility, easy, and gentle construction, as well as versatile and multi-purpose structure regarding their biomedical applications [9]. Binita Chanu et al. investigated anticancer activity of several amino acid-based photo-active monomeric iron(III) with general formula [Fe(L)2], in which Schiff base ligands including (arginine salicylidin, 3,5-di-tert-butyl, benzalidine arginine, and salicylidin Tryptophan) were examined [10]. Kam-Wing Lo and Yin Zhang synthesized a series of iridium complexes with glycine, alanine, and lysine amino acids with [Ir(L)3] structure and its (biological) imaging applications were studied [11].
Chetry et al. studied the synthesis and vibrational spectroscopy of L-Threonine and Cd(L-Threonine)2 complex. The significant role of cadmium’s transition metal ion in cancer diagnosis and preventing the proliferation of tumor cells is undeniable. The L-threonine amino acid is an effective defensive agent against cadmium-induced mortality and cell membrane damage [12]. In this study, chromium (III) metal ion was used to synthesize the primary complex K3 [Cr (O2)4]. In fact, for the first time in our research group, this eight-coordinate chromium (V) complex was used in the synthesis of new chromium complexes. Afterwards, using K3 [Cr (O2)4] complex and tryptophan amino acid, [Cr(Trp)2(OH)(H2O)] complex was synthesized and the results of synthesis of complexes and ligands were identified and investigated.

Figure 1: Structure of tryptophan ligand [13]
Materials and Methods
In this study, chromium complex was synthesized using 2-amino-3-(1-hydrogen-indol-3-yl) propanoic acid (Tryptophan, Trp) ligand and with elemental decomposition identification methods, FT-IR, UV-Vis, conductivity, and melting point were recognized. Likewise, tryptophan, ethanol, potassium hydroxide, potassium chromate, and hydrogen peroxide, made by the German Company “Merck” brand, were used.
Preparation Method of Aqua Based 2-Amino-3-(1-Hydrogen-Indol-3-Yl) Propanoic Acid Hydroxy Chromic Complex (III, [Cr(Trp)2(OH)(H2O)]
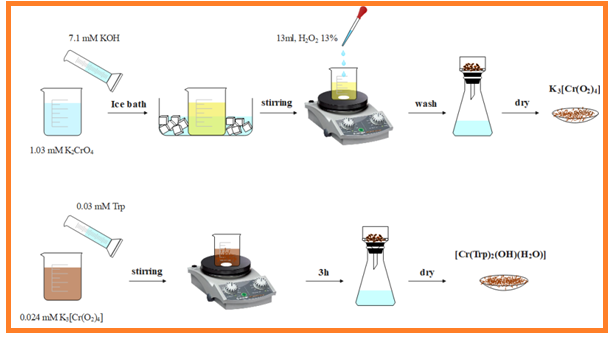
To prepare this complex, the compound K3[Cr(O2)4] was initially synthesized according to the following method [14]: A solution of 2 g (35.64 mmol) of KOH in 5 ml of water was added to a solution of 2 g (10.30 mmol) of K2CrO4 in 10 ml of water and the resulting yellow solution was placed on ice bath until turned into snow, and then while stirring, 13 ml of 13% hydrogen peroxide solution was added drop by drop. After an hour, the resulting brown sediments were filtered, washed using 8 milliliters of ethanol, and then dried at room temperature. The resulting brown residues were used to synthesize the intended complex [11,13]. For the synthesis of [Cr(Trp)2(OH)(H2O)] complex, a solution of 0.1 g (0.49 mmol) of Trp in 15 ml of distilled water, is added to a solution of 0.072 g (0.24 mmol) K3[Cr(O2)4] in 10 mL of water. The resulting light brown mixture is filtered after stirring for 3 hours at room temperature. The complex is collected from the brown sediment after a few days.
mp: 310 °C
Yield: 44% (0.06 g)
Molar conductivity: (1×10-3 mol L-1; EtOH): 24Ω-1cm2 mol-1
Elemental analysis: C22H26CrN4O6 (494.46); Calculated: C, 53.44; H, 5.30; N, 11.33%; Found: C, 52.86; H, 4.70; and N, 12.01%.
IR (KBr disc, cm-1): 3400 s (ν OH), 3051 w (ν NH), 2928-2854 w (νas CH), 2365 w (νs CH), 1612 w (νas COO), 1520 (νs COO), 1454 m (δas CH2), 1385s (δs CH2), and 424 m (Cr-N).
UV-Vis (MeOH, λ max(nm)/ε): 215/41667(π →π*), 345/3250 (n→π*).
Solubility for [Cr(Trp)2(OH)(H2O)]: soluble in (H2O, DMF, MeOH); insoluble in toluene.
Results and Discussion
The [Cr (Trp)2(OH)(H2O)] complex was prepared from the reaction of the Trp ligand with the initial K3[Cr(O2)4] complex.
Identifying aqua bis2-amino-3-(1-hydrogen-indol-3-yl) propanoic acid hydroxychromium complex (III, [Cr (Trp)2(OH)(H2O)]: This complex was formed from the addition of Trp ligand in water to K3[Cr(O2)4] aqueous solution in a ratio of 1:2 (Reaction 1).
Reaction 1: preparation method of [Cr (Trp)2(OH)(H2O)] complex.
The proposed Aqua bis 2-amino-3-(1-hydrogen-indol-3-yl) propanoic acid hydroxochrome (III) complex structure is displayed in Figure 2. This complex melts at 310 °C, and concerning its difference regarding its melting point in comparison to K3[Cr(O2)4], which decomposes at 70 °C, and Trp ligand, which decomposes at 285°C, it is a new compound other than the raw and primary materials. The molar conductivity of the complex in ethanol is 24 Ω-1cm2 mol-1, which, in comparison to the conductivity of the measured salts CuSO4.7H2O (two ions 40), MnCl2.4H2O (three ions 60), and FeCl3.4H2O (three ions 80) in ethanol, is non-electrolyte. Likewise, the elemental analysis of the complex shows that the ratio of ligand to metal is 1:2.

Figure 2: The proposed structure of [Cr(Trp)2(OH)(H2O)] complex
FT-IR
The infrared spectrum is presented below (Figure 3). In the infrared spectrum of the complex [Cr(Trp)2(OH)(H2O)], the vibrational bar corresponding to NH2 (3051 cm-1) has shifted to lower energies by 28 cm-1 compared to the free Trp ligand (3079 cm-1) and the vibration bars of the acetate group (1612 cm-1) have moved to lower energy points by 55 cm-1 compared to the free Trp ligand (1667 cm-1). Furthermore, the Cr-N vibrational bar is present at 424 cm-1, all due to the ligand coordination to chromium metal. The sharp bar corresponding to the vibrations of water molecules and OH- attached to the metal is also observed in the region of 3400 cm-1.

Figure 3: Infrared spectrum: Aquabis 2-amino-3-(1-hydrogen-indol-3-yl)propanoic acid
UV-Vis
In the ultraviolet part of the complex [Cr(Trp)2(OH)(H2O)], two peaks are observed in the electron spectrum (Figure 4). One of the peaks is present in the 215 nm region, and it is attributed to the π→π* transitions of the aromatic ring of this compound [12,15]. It has shifted 13 nm to lower wavelengths (blue shift) compared to Trp free ligand (228 nm). The other peak at 345 nm is attributed to n→ π* transitions of non-bonding electrons of oxygen and nitrogen. This time, compared to the Trp free ligand (361nm), it has shifted 16nm to lower wavelengths (blue shift). These all could be due to the resonance of the aromatic ring with the carbonyl group due to the coordination of the ligand to the central metal, and thus, the peak in the visible region, which is related to metal transitions, has been extended up to the visible part by ligand transitions [16-18].

Figure 4: Electronic spectrum: [Cr(Trp)2(OH)(H2O)]
Conclusion
Concerning the biological importance of amino acids and their complexes with transition elements, “tryptophan” amino acid was used as a ligand, and their chemical structure was studied. To react with chromium using the primary complex K3 [Cr (O2)4] and preparation of the complex [Cr(Trp)2(OH)(H2O)], Ligand 2-amino-3-(1-hydrogen-indol-3-yl) propanoic acid (tryptophan) was used. This compound was identified by elemental analysis, FT-IR, UV-Vis, conductivity, and melting point techniques. For the synthesized complex by similar structures in scientific articles, six-coordinate complexes of chromium were proposed.
Acknowledgments
This study was carried out in Payame Noor University of Tehran, Iran based on the PhD. dissertation. The authors thank the authorities for their cooperation.
Disclosure Statement
No potential conflict of interest was reported by the authors.
Funding
This study did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' Contributions
All authors contributed toward data analysis, drafting and revising the article and agreed to responsible for all the aspects of this work.
Conflict of Interest
The authors declare that there is no conflict of interest in this article.
ORCID
Reihaneh Beiraghi Toosi
https://orcid.org/0009-0005-8338-4867
Mohammad Hakimi
https://orcid.org/0000-0001-8179-1622
Mehdi Dadmehr
https://orcid.org/0000-0002-6016-5988
HOW TO CITE THIS ARTICLE
Reihaneh Beiraghi Toosi, Mohammad Hakimi, Mehdi Dadmehr, Synthesis and Identification of Chromium (III) Complex with 2-Amino-3-(1-hydrogen-indol-3-yl) Propanoic Acid Ligand. Chem. Methodol., 2024, 8(4) 234-240
OPEN ACCESS
©2024 The author(s). This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit: http://creativecommons.org/licenses/by/4.0/
PUBLISHER NOTE
Sami Publishing Company remains neutral concerning jurisdictional claims in published maps and institutional affiliations.
CURRENT PUBLISHER