Document Type : Original Article
Authors
1 Department of Organic Chemistry, Faculty of Chemistry, University of Mazandaran, Babolsar, Iran
2 Department of Analytical Chemistry, Faculty of Chemistry, University of Mazandaran, Babolsar, Iran
Abstract
A novel, high-performance, recyclable heterogeneous catalyst functionalized with melamine was advantageously prepared and rhodanine on magnetic silica was successfully carried out. After that, the Cu ions were coordinated with the existing ligands decorated on the magnetic silica surface. This catalyst was identified by different analyses and acted as a recoverable catalyst for xanthene synthesis. Its advantages include the solvent-free conditions of the reaction, the high efficiency of the products in a short period of time, as well as its simple collection from the reaction environment. Moreover, the prepared sorbent has been developed for the extraction of diazinon from different real samples through magnetic solid phase extraction (MSPE). Under optimal conditions, the linear range for measuring diazinon was 0.5-200 µg L-1. The coefficient of determination (R2) was 0.9985. The limit of detection (LOD) of the method was 0.08 μg L-1. Relative standard deviation values (RSD%) for the concentration of 10.0 μg L-1 and 100 μg L-1 were between 5.3% and 5.9%. Finally, the proposed method was used to determine diazinon in orange and agricultural water. The results showed that the percentage of relative recovery of the selected toxin in the studied real samples was between 92 and 105.
Moreover, the prepared sorbent has been developed for the extraction of diazinon from different real samples through magnetic solid phase extraction (MSPE). The parameters affecting the extraction were optimized by response surface methodology (RSM). The optimal extraction conditions were 15 min for contact time, 7 mg for sorbent amount, and 0% (w/v) for NaCl concentration.
Moreover, 20 µL of methanol was used to elute the extracted analyte. Under optimal conditions, the linear range for measuring diazinon was 0.5-200 µg L-1. The coefficient of determination (R2) was 0.9985. The limit of detection (LOD) of the method was 0.08 μg L-1. Relative standard deviation values (RSD%) for the concentration of 10.0 μg L-1 and 100 μg L-1 were between 5.3% and 5.9%. Finally, the proposed method was used to determine diazinon in orange and agricultural water. The results showed that the percentage of relative recovery of the selected toxin in the studied real samples was between 92 and 105.
Graphical Abstract
Keywords
Main Subjects
Introduction
Heterocyclic compounds are a clear example of chemical structures that have important roles and functions in human life. In addition to carbon, this category of organic compounds has heteroatoms instance as nitrogen, oxygen, or sulfur. Heterocyclic compounds have wide and vital applications in various industries and biological fields. Among its applications in industries, we can mention the production of paints, antioxidants, antimicrobial herbicides, corrosion inhibitors, copolymers, and agriculture. They are also mainly in the structure of many drugs such as penicillin, and cephalosporin; There are alkaloids like vinblastine, morphine, reserpine, etc. [1-4].
Xanthene and benzoxanthene are an important group of three-ring heterocycles containing oxygen and are biologically active. They are an important group of compounds with different uses for example anti-tumor, anti-viral, anti-inflammatory activities, food additives, and anti-fungal [5-9].
In the past years, the study of rhodanine and its derivatives, which are a main group of heterocyclic structures, has been of particular importance. Rhodanines are present in the chemical skeleton of drugs like the behavior toward type II diabetes, Alzheimer's disease, cancer, and cataracts. These compounds have antibacterial, antifungal, antiviral, antimalarial, insecticide and herbicide activities [10-13].
In recent decades, the use of nanomaterials, especially magnetic nanoparticles, has become very popular in various fields such as drug delivery. Magnetic nanoparticles are of interest owing to their unique characteristics like easy separation, high recyclability, large surface area, proper dispersion, and low toxicity [14-19].
Many coating materials such as noble metals, polymers, and silica have been used for magnetic nanoparticles. Among the various coatings of biocompatible and hydrophilic silica material due to the preservation of nanoparticles against various environmental factors for example changes in pH and air, as well as the presence of silanol groups on its surface, which easily connects with molecules and ligands, is more important, and can be used in biomedical fields [20,21].
Copper (Cu) nanoparticles are cheap, have good efficiency, low catalyst loading, and short reaction time among their advantages. They are used in various fields such as water purification, heat transfer systems, and biomedicine, and also have antibacterial activity. To overcome the problems that limit the activity of catalysts such as aggregation, oxidation, and difficult separation, they are placed on various substrates like silica, organometallic frameworks, polymers, and zeolites [21,22].
In this way, in continuation of the past catalytic works [23,29] and to develop a new practical method, we designed a green novel catalyst system on a magnetic silica substrate based on melamine and rhodanine. Then, this heterogeneous catalyst (Fe3O4@SiO2@Mel-Rh-Cu) was used in the synthesis reaction of xanthenes, which are heterocyclic compounds. In the next part of this study, the prepared sorbent was used for the MSPE of diazinon The Box-Behnken Design (BBD) and Response Surface Methodology (RSM) were used to optimize the practical extraction factors after they had been tested with the Plackett-Burman Design (PBD). Pesticides are widely used in the cultivation of plants and plant products to prevent and control diseases and insect pests. Pesticides include insecticides, fungicides, herbicides, or a combination of one or more types of chemicals that are used primarily in agriculture to kill pests. Toxin concentrations in plants and agricultural products should be measured. Due to the presence of pesticides and their high toxicity, it is nowadays important to develop and extend methods to determine the amount of pesticide residues in fruit and water samples. The toxic effects of diazinon are due to inhibition of acetylcholinesterase. Diazinon is an insecticide, and one of its detection methods is chromatography, which offers a low level of detection but requires the pretreatment of samples before analysis. Therefore, it is important to provide an affordable and reliable method to measure toxins.
Here, the magnetic sorbent was used for the presentation of a novel extraction method. The obtained results showed good extraction efficiency, no memory effect, high adsorption capacity, and good reproducibility. Therefore, the desired adsorbent can be used to extract different analytes in real samples.
Experimental
Each of the solvents or materials is from Merck and Fulica. NMR (1H and 13C) spectra were measured with a Bruker DRX-400 AVANCE spectrometer. Melting points were calculated using the Electrothermal IA9100 (Essex, UK). XRD was performed using the PHILIPS, PW1730 instrument. TGA was performed with a Netzsch device (Selb, Germany). The SEM and VSM analyses were performed using TESCAN MIRA III and MDKB Kashan Desert Magnet Company. The power of the external magnet is 1.2 T.
Synthesis of Fe3O4@SiO2
Magnetic nanoparticles were synthesized according to the published literature [21]. In the next step, Stober's method was used to synthesize Fe3O4@SiO2. 1 g Fe3O4 in ethanol (100 mL), and deionized water (50 mL) was sonicated (40 min), and then 25% ammonia was added to the reaction (4 mL). TEOS (Tetraethyl orthosilicate) (4 mL, 3.73 g) was then added dropwise and stirred for 18 h. The Fe3O4@SiO2 was collected by a magnetic field, washed with water/ethanol, and dried.
Synthesis of Fe3O4@SiO2@CPTMS
Initially, 1 g Fe3O4@SiO2 was dispersed in dry toluene for 35 min. 1 mL (1.09 g) 3-chloropropyltrimethoxysilane (CPTMS) was added under reflux conditions and N2 atmosphere. After 18 h, the reaction mixture was filtered and washed with toluene [30].
Synthesis of Fe3O4@SiO2@Mel
1 g of Fe3O4@SiO2@CPTMS was dispersed in 50 ml of ethanol. Then 2 mmol of melamine was added and refluxed for 22 h. The prepared Fe3O4@SiO2@Melamine (Fe3O4@SiO2@Mel) was washed with ethanol/water and dried [31].
Synthesis of Fe3O4@SiO2@Mel-Rh
Initially, 1 g of Fe3O4@SiO2@Mel in 30 ml of dry toluene was placed under ultrasonic radiation for 30 min to disperse it well. Then 3 ml (3.48 g) of 1, 4-dichlorobutane and 2 mmol (0.33 g) of KI were added to the solution, it was kept for 18 h under reflux and nitrogen conditions. The resulting mixture was separated from the solvent with the help of a magnet, washed multiple times with ethyl acetate, and dried at 60 °C [30]. In the next step, 1 g of synthetic material from the former stage was dispersed in DMF for 30 min. 3 mmol (0.4 g) of rhodamine, was added and stirred at 90 °C for 30 h. Fe3O4@SiO2@Melamine-rhodanine (Fe3O4@SiO2@Mel-Rh) was washed with water after separation [30].
Synthesis of Fe3O4@SiO2@Mel-Rh-Cu
1 g of Fe3O4@SiO2@Mel-Rh was dispersed in water by ultrasonic waves, and then 0.1 g Cu(OAc)2 was added and stirred for 15 h at 80 °C. The resulting was separated by a magnetic field and washed with deionized water (Scheme 1).
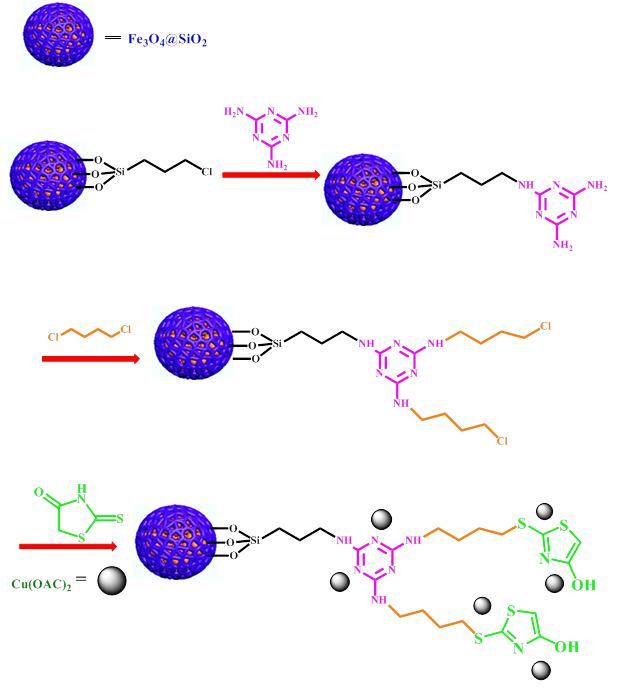
Scheme 1: Catalyst synthesis steps Fe3O4@SiO2@Mel-Rh-Cu
General Procedure for the Synthesis of Xanthenes
The arylaldehyde (1 mmol), dimedone (1 mmol, 0.14 g), and 2-naphthol (1 mmol, 0.14 g) were mixed together in the presence of Fe3O4@SiO2@Mel-Rh-Cu catalyst under solvent-free conditions at 50 °C. Finally, ethanol was added to the reaction mixture and the Fe3O4@SiO2@Mel-Rh-Cu was separated using a magnetic field. The resulting precipitate was recrystallized in ethanol (Scheme 2).
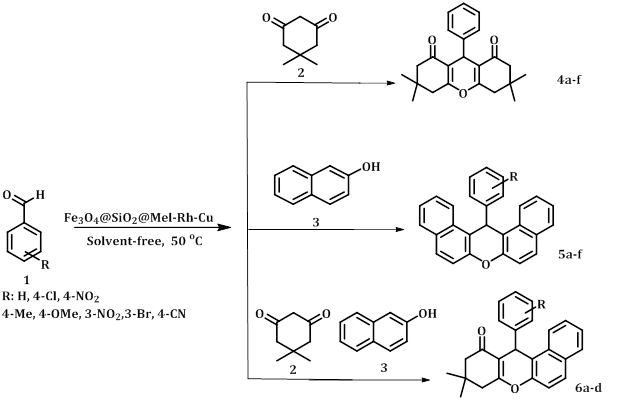
Scheme 2: Synthesis of xanthenes using Fe3O4@SiO2@Mel-Rh-Cu
Selected Spectral Data
3,3,6,6-Tetramethyl-9-phenyl-3,4,5,6,7,9-hexahydro-1H-xanthene-1,8(2H)-dione (4a):
1H-NMR (CDCl3, 400 MHz): δ 1.03 (s, 6H), 1.14 (s, 6H), 2.20 and 2.26 (4H, J=16.0 Hz,), 2.50 (s, 4H), 4.75 (s, 1H), and 7.20–7.27 (m, 4H). 13C-NMR (CDCl3, 100 MHz): δ 26.4, 30.0, 31.5, 33.4, 39.9, 49.5, 114.6, 129.0, 130.0, 133.1, 143.4, 163.5, and 197.1.
9-(4-Methoxyphenyl)-3,3,6,6-tetramethyl-3,4,5,6,7,9-hexahydro-1H-xanthene-1,8(2H)-dione (4d):
1 H-NMR (CDCl3, 400 MHz): δ 1.00 (s, 6H), 1.15 (s, 6H), 2.17 and 2.30 (4H, CH2), 2.51 (s, 4H), 4.85 (s, 1H), 7.50 (d, 2H), and 8.15 (d, 2H). 13C NMR (CDCl3, 100 MHz): δ 26.9, 30.0, 31.3, 33.6, 41.0, 51.0, 111.0, 115.1, 123.5, 130.1, 147.4, 151.5, 163.0, and 196.2.
14-Phenyl-14H-dibenzo[a,j]xanthene (5a):
δ 1H-NMR (CDCl3, 400 MHz): δ 6.50 (s, 1H), 7.13 (d, 2H), 7.43–7.52 (m, 6H), 7.60–7.64 (m, 2H), 7.83 (d, 2H), 8.56 (d, 2H), and 8.34 (d, 2H). 13C-NMR (CDCl3, 100 MHz): δ 38.4, 117.8, 119.2, 121.5, 125.4, 127.9, 128.8, 129.0, 129.1, 129.5, 131.1, 131.3, 132.1, 143.5, and 148.7.
9‑Dimethyl‑12‑(4‑nitrophenyl) ‑9,10‑dihydro‑8H‑benzo[a]xanthen‑11(12H) ‑one (6c) :
1H-NMR (CDCl3, 400 MHz): δ 0.97 (s, 3H, CH3), 1.16 (s, 3H, CH3), 2.25 and 2.38 (2H, , CH2), 2.62 (s, 2H, CH2), 5.84 (s, 1H, CH), 7.38 (d, 1H, CH), 7.40-7.48 (m, 2H, CH), 7.54 (d, 2H, CH), 7.81-7.86 (m, 3H, CH), and 8.05 (d, 2H, CH). 13C-NMR (CDCl3, 100 MHz): δ 27.0, 29.3, 32.2, 34.9, 41.4, 50.7, 112.9, 116.0, 117.1, 123.1, 123.6, 125.2, 127.3, 128.6, 129.3, 129.6, 131.0, 131.5, 146.3, 151.8, 164.6, and 196.7.
12-(3-Bromophenyl)-9,9-dimethyl-9,10-dihydro-8H-benzo[a]xanthen-11(12H)-one (6d):
1H-NMR (CDCl3, 400 MHz): δ 0.98 (s, 3H), 1.17 (s, 3H), 2.25 and 2.38 (2H, CH2), 2.73 (s, 2H), 5.78 (s, 1H), 7.39 (d, 1H), 7.41-7.50 (m, 2H), 7.59 (d, 2H), 7.83-7.88 (m, 3H), 8.08 (d, 2H). 13C-NMR (CDCl3, 100 MHz): δ 26.9, 30.0, 33.2, 35.0, 41.45, 51.0, 113.0, 116.1, 117.2, 123.2, 123.8, 125.6, 127.5, 128.9, 129.8, 129.3, 131.1, 131.3, 146.7, 151.9, 164.2, and 196.8.
Results and Discussion
We report in this research the synthesis and characterization of a novel heterogeneous catalyst of Cu(OAc)2 immobilized on a magnetic silica substrate. For this purpose, magnetic silica was first prepared using literature, and then 3-chloropropyltrimethoxysilane was used to connect the substrate with melamine. The next step involves the grafting of melamine to rhodanine via a 1,4-dichlorobutane as the linker. Subsequently, in the final step, copper (II) acetate was utilized for the metal coordination complex to provide Fe3O4@SiO2@Mel-Rh-Cu.
Characterization of Catalyst
After the synthesis of the catalyst, FT-IR TGA, XRD, FESEM, EDX, elemental mapping, VSM, and ICP techniques were used to identify it.
In the FT-IR spectrum, the peak in the regions of 590, 956, and 1063 cm-1 is associated with the stretching vibrations of Fe-O, Si-O-Fe, and Si-O bonds, respectively. C-H stretching vibrations appear in the range of 2922-2983 cm-1. The peak in the range of 1230 cm-1 refers to CH2-Cl bending (in the spectrum of Fe3O4@SiO2@CPTMS). The presence of peaks in the 1548 and 1650 cm-1 regions is attributed to the stretching vibrations of C=C, C=N, and the characteristic peaks at 3338-3469 cm-1 are also associated with NH and NH2 groups, which indicates the successful connection of the melamine ring to the substrate (in the spectrums of melamine, and Fe3O4@SiO2@Mel@Cl (Figure 1).
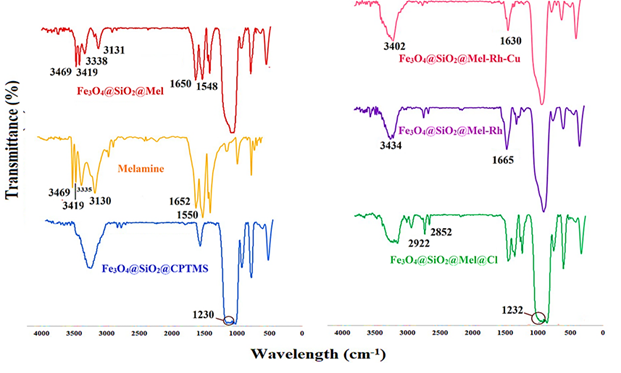
Figure 1: FTIR spectrum of Fe3O4@SiO2@Mel-Rh-Cu synthesis steps
The increase in the height of the peak in the region of 2852-2922 cm-1 and the peak in the region 1232 cm-1 corresponding to C–H wag (–CH2-Cl) stretching presence of alkyl halides (in the spectrum of Fe3O4@SiO2@Mel@Cl) indicates the binding of 1,4-dichlorobutane to melamine. C-N stretching bond of rhodanine appeared at 1665 cm-1 and OH stretching vibrations were also attributed to 3434 cm-1 (in the spectrum of Fe3O4@SiO2@Mel-Rh-Cu), which proof of rhodanine is binding. FT-IR spectrum was analyzed to confirm the metallization of Fe3O4@SiO2@Mel-Rh. The change in curves Fe3O4@SiO2@Mel-Rh-Cu from 1665 to 1630 cm-1 and 3433 to 3402 cm-1 associated with C-N and OH stretching bond is attributed to coordination with metal.
TGA curve is used to determine the percentage of supported organic functional groups on the Fe3O4@SiO2@Mel-Rh-Cu catalyst surface in Figure 2. Weight loss at temperatures below 200 C is related to the removal of surface water. 20 % weight loss at 300 to 800 C is associated with the decomposition of organic groups placed on the Fe3O4@SiO2 substrate (Figure 2).

Figure 2: TGA curve of Fe3O4@SiO2@Mel-Rh-Cu
Through XRD analysis of Fe3O4, Fe3O4@SiO2@Mel-Rh, and Fe3O4@SiO2@Mel-Rh-Cu, their crystal structure was investigated. The XRD patterns of all three compounds show six diffraction peaks related to the inverse cubic spinel structure of Fe3O4 at 2θ: 30.26° (220), 35.86° (311), 43.25° (400), 53.22° (422), and 57.01° (511) which indicates the preservation of the crystal structure of the composition after modification. Due to the dispersion of Cu nanoparticles in the matrix, which cannot form a regular crystal lattice, no diffraction peak for Cu nanoparticles was observed for Fe3O4@SiO2@Mel-Rh-Cu.The immobilization of Cu species was confirmed by IR, ICP-OES and EDX analysis (Figure 3).
The morphology of the synthesized nanocatalyst was considered by SEM analysis. FE-SEM images of Fe3O4 show the spherical shape of nanoparticles with a size of 8 nm. After placing the silica layer, the organic ligands of the catalyst morphology were preserved and the diameter of the Fe3O4@SiO2@Mel-Rh-Cu nanoparticles increased to an average of 23 nm (Figure 4).

Figure 3: XRD patterns of Fe3O4, Fe3O4@SiO2@Mel-Rh and, Fe3O4@SiO2@Mel-Rh-Cu

Figure 4: SEM analysis of Fe3O4 and Fe3O4@SiO2@Mel-Rh-Cu
The EDX analysis confirmed the elements Fe, Si, O, C, S, and N (Figure 5). The analysis of the element distribution also shows a uniform distribution of these elements on the surface of the Fe3O4@SiO2@Mel-Rh-Cu (Figure 6).
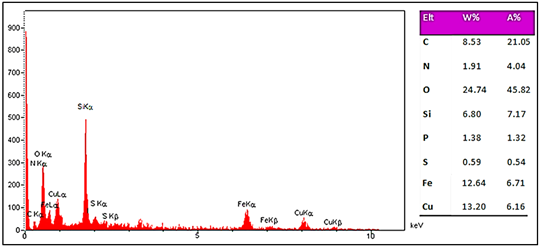
Figure 5: EDx pattern of Fe3O4@SiO2@Mel-Rh-Cu

Figure 6: Elemental mapping of Fe3O4@SiO2@Mel-Rh-Cu
Analysis of the magnetic properties of Fe3O4 and Fe3O4@SiO2@Mel-Rh-Cu was done by VSM analysis. The magnetic curve for Fe3O4 and Fe3O4@SiO2@Mel-Rh-Cu showed 65 and 47 emu/g, respectively. The saturation magnetization of the Fe3O4@SiO2@Mel-Rh-Cu has decreased due to the silica coating and the diamagnetic contribution of the added organic parts. However, the magnetic property of the catalyst is still well preserved and it is easily separated from the solution (Figure 7).

Figure 7: Magnetization curves of (a) Fe3O4 and (b) Fe3O4@SiO2@Mel-Rh-Cu
Finally, based on ICP analysis, the exact loading of Cu on the surface of the synthesized catalyst was found to be 2.96 mmol/g.
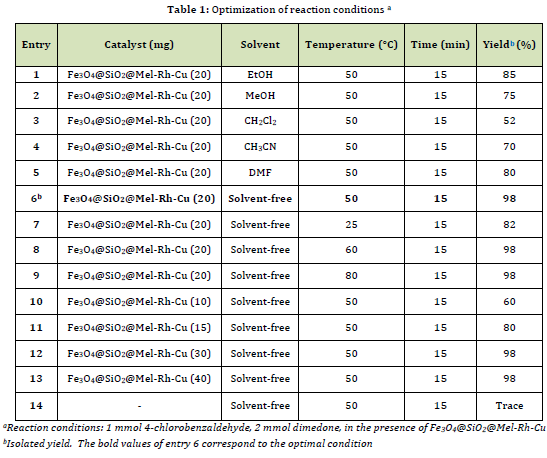
To illustrate the power and efficiency of the synthesized catalyst, we measured its activity in the synthetic reaction between aromatic aldehydes, dimedone, or 2-naphthol. For this purpose, the reaction between 4-chlorobenzaldehyde (1 mmol, 0.14 g) and dimedone (2 mmol, 0.14 g) was selected as the model until the optimal conditions were reached.
Initially, the reaction was accomplished in solvents for instance ethanol, methanol, dichloromethane, acetonitrile, dimethylformamide, and solvent-free conditions. It was observed that solvent-free conditions are the most suitable choice for this catalyst in the reaction (Table 1, entries 1-6). The reaction efficiency was also evaluated in temperature conditions (25 to 80 C). The observations indicated that increasing the temperature by more than 50 C did not influence the reaction yield (Table 1, entries 6-9). The best amount of catalyst (estimated between 10-40 mg) was calculated as 20 mg (Table 1, entries 6 and 10-13). Finally, without a catalyst, the reaction was performed, which showed a trace yield of the product (Table 1, entry 14).
Investigating and Optimizing the Type of Desorption Solvent
The choice of the type of solvent and adsorption is a very effective parameter for the extraction process. A suitable solvent can desorb the adsorbed analytes with the smallest volume from the surface of the adsorbent. In this study, the solvents acetonitrile, methanol, ethanol, isopropanol and ethyl acetate were selected for the desorption process. The results showed that methanol exhibited better efficiency than the other solvents used. Therefore, methanol was chosen as the most suitable desorption solvent (Figure 8).

Figure 8: The effect of type of desorption solvent on magnetic solid phase microextraction process
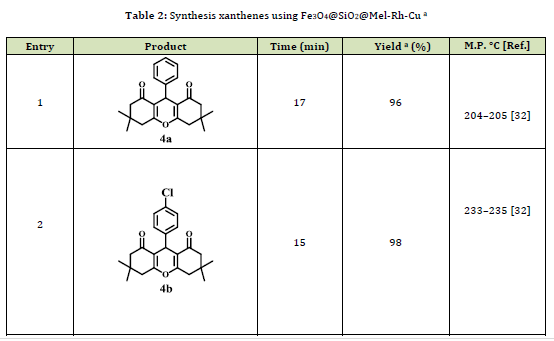
After determining the optimal conditions, to expand the reaction range, different aromatic aldehydes with dimedone (2) and/or 2-naphthol (3) were used in the presence of Fe3O4@SiO2@Mel-Rh-Cu (Table 2). The outcomes are displayed in the electron-withdrawing groups unlike electron-donating groups will increase the efficiency and decrease the reaction time.



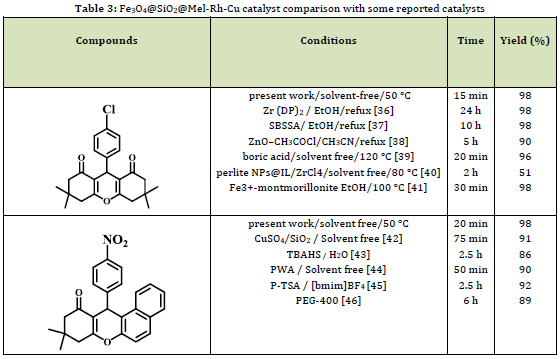
In the next step, the performance of the synthetic catalyst was compared with the previously published results (Table 3). The technique used is simpler, easier to separate, more effective, and with higher yields in less time for this reaction than other methods.
Synthesis Mechanism of Xanthene Derivatives by Fe3O4@SiO2@Mel-Rh-Cu
At the beginning of the reaction, the carbonyl group of aldehydes is activated by the catalyst so that the nucleophilic attack can be carried out more easily. Intermediate A is formed by the attack of dimedone on aldehyde, and then by adding another molecule of dimedone to that intermediate B, and finally by removing the water molecule, the desired product is created (Scheme 3).
One of the important processes in the synthesis of catalysts is their recovery and reusability. To check this, the reaction of the model was used. Subsequent to the ending of the reaction, the Fe3O4@SiO2@Mel-Rh-Cu was collected by the magnetic field and washed with ethanol and after drying, it was used in a further step. This catalyst can be reused for at least 7 steps. Likewise, after seven consecutive cycles of reuse of Fe3O4@SiO2@Mel-Rh-Cu, the XRD patterns, and FT-IR spectra do not indicate remarkable differences with the fresh figure, and the properties and activity of the catalyst remain unchanged (Figure 9).
Preparation of Real Samples
The purpose of this study was to investigate the ability of the prepared adsorbent in to measure the real samples of oranges and agricultural waste water. The desired sample was bought from a local market and after washing with water, it was peeled and crushed completely. To provide the samples, 10 g of it was crushed and added to 15 ml of acetonitrile and 5 ml of water. Further, 1 g of NaCl was added to the solution and the resulting mixture was ultrasonicated (min). Finally, 4 g of MgSO4 was added to the mixture and the mixture was centrifuged for 5 min. Thereafter, 5 mL of the supernatant was removed and centrifuged again for (3 min). The resulting solution was used to extract target analytes. The standard addition method was used for quantifying tasks. For this purpose, standard concentrations of 2, 5, and 10 μg L-1 of target analytes were added to the samples. After the extraction process, finally, the extracted eluents were injected into HPLC-UV and the concentration of diazinon in the real samples was determined.
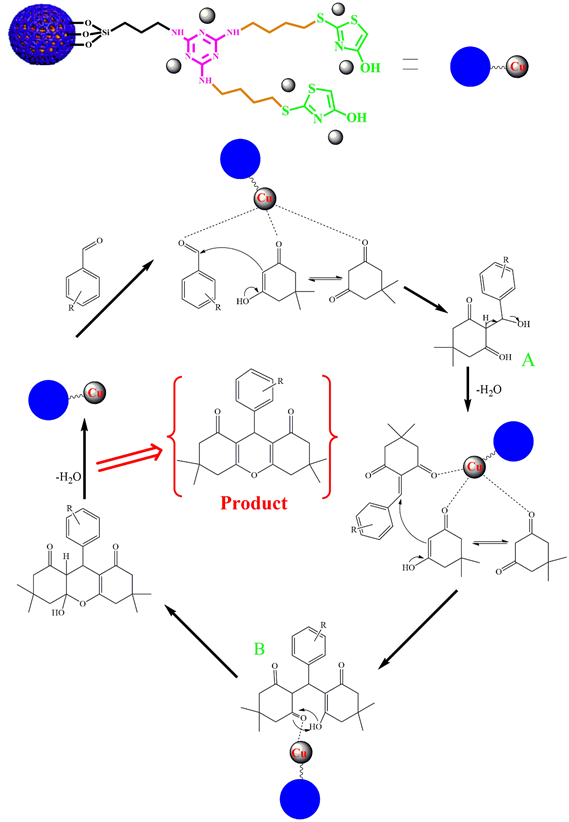
Scheme 3: Synthesis mechanism of xanthene derivatives by Fe3O4@SiO2@Mel-Rh-Cu

Figure 9: (A) Catalyst reusability diagram of the model reaction, (B) IR spectra of Fe3O4@SiO2@Mel-Rh-Cu reused after seven cycles, and (C) XRD patterns of Fe3O4@SiO2@Mel-Rh-Cu reused after eleven cycles
Instrumentation
Separation, detection and quantification of the extracted analyte was performed using a Waters HPLC instrument equipped with a dual UV-Vis detector model 2487 (Waters Assoc, Milford, MA, USA), a binary solvent pump model 1525 and an injection valve (Rheodyne 7725i (Cotati, CA, USA)) containing a 20-μL sample loop. Separation of the selected analytes was performed at room temperature on a Waters Symmetry ® reversed phase C18 column (25 mm × 4.6 mm I.D., 5 μm particle size). The analyte was eluted by an isocratic program at the flow rate of 1.0 mL min−1. The mobile phase consisted of acetonitrile and water (80:20). The UV detector was set at 239 nm. Diazinon, was eluted at 6.1 min.
MSPE Procedure
To investigate the performance of the MSPE method (Magnetic solid-phase extraction) based on the prepared sorbent for diazinon extraction, the working solution of diazinon (100 µL-1) was prepared and used. The initial step was screening using one factor at a time methodology and the second was optimization by Box-Behnken experimental design (BBD) to investigate optimal extraction conditions. Therefore, after examining the factors affecting the extraction process, the best condition of extraction was achieved. The exact values of each of the parameters depend on the values determined by the BBD table, but in general, the procedure was as follows:
10 ml of the sample containing the target analyte was introduced into the extraction tube (pH=7). A precise amount of adsorbent (7 mg) was subsequently added to the tube and thoroughly mixed by sonication for 15 min. The mixture was then subjected to an external magnet. The supernatant was decanted. At this juncture, an appropriate volume of desorption solvent (25 µL of methanol), as per the BBD table, was added to the precipitated adsorbents and sonicated for 1 min. For chemical analysis, 20 µL of the resultant mixture was injected into the HPLC-UV.
Design of Experiments And Optimizing (DOE)
In the next step, the mathematical modeling and assessment of responses were conducted using the Minitab software. RSM optimization (Response surface methodology) is a key application of DOE. Various design techniques have been introduced in this method, and in this study, due to the extensive number of tests required and the potential interactions between the parameters, the BBD (Box-Behnken Design) was employed to identify the interferences among them and determine the optimal levels of these variables.
BBD enables the estimation of model parameters and necessitates fewer trials in the case of three variables. As such, five quantitative variables, namely the amount of sorbent, extraction time, ionic strength, desorption time, and eluent volume were estimated, and 46 runs were randomly designed based on them. Three-dimensional surface plots or contour plots were used to graphically display the relationship and interaction between the dependent variables and the absorption as the response.
Method Evaluation
To estimate the validity of the proposed technique, the performance metrics, the limit of detection (LOD), the linear dynamic range (LDR), the limit of quantification (LOQ), the enrichment factor (EF), and the relative standard deviations (RSD), were calculated. The target analyte concentration range is called LDR, where the amount of signal is proportional to the analyte concentration. A series of 9 extraction experiments were done with 9 different concentrations of 0.5, 1, 2, 5, 10, 20, 50, 100, and 200 μg L-1 of the chosen analyte in DI water. The linear range of diazinon was obtained in 0.5-200 μg L-1. The coefficients of determination (R2) for diazinon were 0.9985. The LODs and LOQs were determined based on the S/N=3 and 10, respectively. The limit of detection (LOD) of the technique for diazinon was 0.08 μg L-1. Relative standard deviation (RSD %) values for having 10 μg L-1, 50 μg L-1, and 100 μg L-1 were also obtained between 5.3% and 6.2%. The following equation (EF: The enrichment factor) is used to calculate the concentration of the extracted analyte in the optimal eluent and the primary concentration of the target analyte (Equation (1)):
![]()
The estimated EF was 69. The mechanism for the extraction of the target analyte from the sample solution to the sorbent might be owing to the π-π stacking interaction, hydrogen bonding, and Van der Waals interaction between selected pesticides and the synthesized sorbent.
Real Sample Analysis
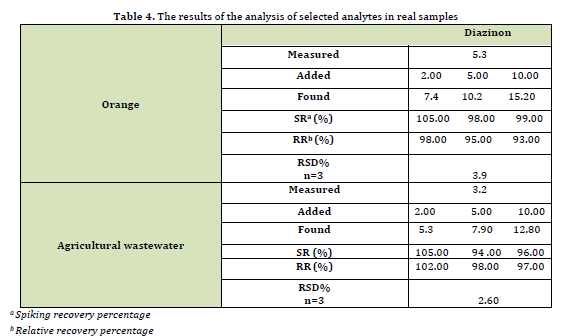
To evaluate the accuracy and capability of the technique, real sample analysis was used. For the quantitative determination of analytes, the addition technique was used as a standard. In order to accomplish this, three different concentrations (2, 5, and 10 μg/liter) were added to the samples and each concentration was replicated three times (n = 3). Also, relative recovery and spike recovery were calculated. Relative recovery (RR) was defined as the ratio of the distinguished analyte in real samples and pure water samples spiked with the same amount of analyte were measured for concentration. Spiking recovery was calculated using the following equation:
![]()
Where,
Cfound : the concentration after MSPE
Creal: the spiked concentration into the real sample
Cadded: the concentrations of the analyte (µg L-1) in the real sample
Also, by repeating the test three times, the value of RSD% was obtained (Table 4). According to the obtained data, it can be confirmed that the above technique is suitable for the desired analyte in real samples.
Conclusion
In this study, we have fabricated a new catalyst from rhodanine (whose derivatives have medicinal properties), melamine linker (which is utilized in the polymer industry), and then immobilized inexpensive copper metal ions on it. The Fe3O4@SiO2@Mel-Rh-Cu was distinguished by some techniques like FT-IR, TGA, XRD, VSM, FE-SEM, EDX, and ICP. The prepared Fe3O4@SiO2@Mel-Rh-Cu can be exploited as a remarkable, novel, and reusable catalyst for Xanthene synthesis. Among the advantages of the synthesized catalyst, we can mention the green conditions (without solvents) of the reaction, the high efficiency of the products in a short period, and suitable temperature conditions. Furthermore, it can be easily collected from the reaction environment and reused up to seven times.
The prepared sorbent was also used for the MSPE of diazinon followed by quantification through HPLC-UV. The proposed technique has the capability of separation and preconcentration in one step. Here, the extraction efficiency of the technique was improved by designing an optimization experiment of important and influencing variables, and then the figure of merit of the method was calculated. Furthermore, no matrix effect was observed with this technique. In summary, the results presented in the manuscript show that the proposed technique is satisfactory for the designation of small amounts quantities amounts of toxins in real samples.
Acknowledgments
This study was supported by the research grant of the University of Mazandaran (No. 33/38554)
ORCID
Sahar Peiman
https://orcid.org/0009-0000-7509-9277
Behrooz Maleki
https://orcid.org/0000-0002-0322-6991
Milad Ghani
https://orcid.org/0000-0003-1210-1856
HOW TO CITE THIS ARTICLE
Peiman, B. Maleki, M. Ghani. Fe3O4@SiO2@Mel-Rh-Cu: A High-Performance, Green Catalyst for Efficient Xanthene Synthesis and Its Application for Magnetic Solid Phase Extraction of Diazinon Followed by Its Determination through HPLC-UV. Chem. Methodol., 2024, 8(4) 257-279
OPEN ACCESS
©2023 The author(s). This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit: http://creativecommons.org/licenses/by/4.0/
PUBLISHER NOTE
Sami Publishing Company remains neutral concerning jurisdictional claims in published maps and institutional affiliations.
CURRENT PUBLISHER


